 Limited Edition Golden Llama is here! Check out how you can get one.
Limited Edition Golden Llama is here! Check out how you can get one.  Limited Edition Golden Llama is here! Check out how you can get one.
Limited Edition Golden Llama is here! Check out how you can get one.
 Offering SPR-BLI Services - Proteins provided for free!
Offering SPR-BLI Services - Proteins provided for free!  Offering SPR-BLI Services - Proteins provided for free!
Offering SPR-BLI Services - Proteins provided for free!
 Here come GMP Grade Cytokines!Free Sample is available!
Here come GMP Grade Cytokines!Free Sample is available!  Here come GMP Grade Cytokines!Free Sample is available!
Here come GMP Grade Cytokines!Free Sample is available!
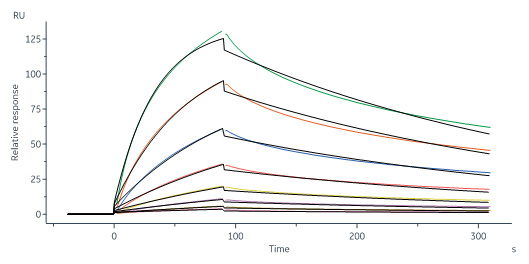
MERS S1 protein, His Tag (Cat. No. S1N-M52H5) immobilized on CM5 Chip can bind Cynomolgus DPPIV, His Tag (Cat. No. DP4-C52H9) with an affinity constant of 60.7 nM as determined in a SPR assay (Biacore 8K) (Routinely tested).
| Name | Research Code | Research Phase | Company | First Brand Name | First Approved Country | First Indication | First Approved Company | First Approved Date | Indications | Clinical Trials |
|---|---|---|---|---|---|---|---|---|---|---|
| Gemigliptin L-tartrate Sesquihydrate | LC-150444; LC-15-0444 | Approved | Lg Life Sciences Ltd | Gemiglo, Zemiglo | South Korea | Diabetes Mellitus, Type 2 | Lg Life Sciences Ltd | 2012-06-27 | Diabetes Mellitus, Type 2; Hepatic Insufficiency | Details |
| Gemigliptin L-tartrate Sesquihydrate/Metformin Hydrochloride | Approved | Lg Life Sciences Ltd | ZemiMet SR | South Korea | Diabetes Mellitus, Type 2 | Lg Life Sciences Ltd | 2015-01-01 | Diabetes Mellitus, Type 2 | Details | |
| Teneligliptin Hydrobromide Hydrate/Metformin Hydrochloride | Approved | Handok Inc | Tenelia M SR | South Korea | Diabetes Mellitus, Type 2 | Handok Inc | 2015-11-03 | Diabetes Mellitus, Type 2 | Details | |
| Evogliptin | DA-1229 | Approved | Dong-A Pharmaceutical Co Ltd | Suganon, Sugarnon | South Korea | Diabetes Mellitus, Type 2 | Dong-A Pharmaceutical Co Ltd | 2015-10-02 | Diabetes Mellitus, Type 2; Kidney Diseases; Aortic valve diseases; Liver Diseases | Details |
| Saxagliptin Hydrate | BMS-477118; BMS-477118-11; OPC-262 | Approved | Bristol-Myers Squibb Company | 安立泽, Onglyza | EU | Diabetes Mellitus, Type 2 | Astrazeneca Ab | 2009-07-31 | Diabetes Mellitus, Type 1; Diabetes Mellitus, Type 2; Hypoglycemia; Diabetes Mellitus | Details |
| Teneligliptin Hydrobromide Hydrate | MP-513 | Approved | Mitsubishi Tanabe Pharma | Tenelia, Tenglyn | Japan | Diabetes Mellitus, Type 2 | Mitsubishi Tanabe Pharma, Daiichi Sankyo Co Ltd | 2012-06-29 | Diabetes Mellitus, Type 2 | Details |
| Retagliptin Phosphate | SP-2086 Phosphate | Approved | Jiangsu Hengrui Medicine Co Ltd | 瑞泽唐 | Mainland China | Diabetes Mellitus, Type 2 | Jiangsu Hengrui Medicine Co Ltd | 2023-06-27 | Diabetes Mellitus, Type 2; Renal Insufficiency; Diabetes Mellitus | Details |
| Sitagliptin Phosphate Monohydrate/Ipragliflozin L-Proline | MK-0431J | Approved | Merck & Co Inc, Astellas Pharma Inc | Sujanu | Japan | Diabetes Mellitus, Type 2 | null | 2018-03-23 | Diabetes Mellitus, Type 2 | Details |
| Linagliptin | BI-1356; BI-1356-BS | Approved | Boehringer Ingelheim Gmbh | 欧唐宁, Ondero, Trajenta, Trayenta, Trazenta, Tradjenta | United States | Diabetes Mellitus, Type 2 | Boehringer Ingelheim Gmbh | 2011-05-02 | Prediabetic State; Diabetes Mellitus, Type 2; Albuminuria; Renal Insufficiency; Schizophrenia; Respiratory Tract Diseases; Coronavirus Disease 2019 (COVID-19); Hypertension; Endocrine System Diseases; Coronavirus Infections; Metabolic Diseases; Hepatic Insufficiency; Glucose Metabolism Disorders; Inflammation; Diabetes Mellitus; Ventricular Dysfunction, Left | Details |
| Rosuvastatin Calcium/Gemigliptin L-tartrate Sesquihydrate | Approved | Lg Life Sciences Ltd | ZemiStatin | Diabetes Mellitus, Type 2; Dyslipidemias | Details | |||||
| Alogliptin Benzoate | SYR-322; TAK-322 | Approved | Takeda | Nesina, 尼欣那, Vipidia | Japan | Diabetes Mellitus, Type 2 | Takeda | 2010-04-06 | Diabetes Mellitus, Type 2; Acute Coronary Syndrome; Diabetes Mellitus | Details |
| Empagliflozin/Linagliptin/Metformin | Approved | Boehringer Ingelheim Gmbh, Eli Lilly And Company | Trijardy XR | United States | Diabetes Mellitus, Type 2 | Boehringer Ingelheim Gmbh | 2020-01-27 | Diabetes Mellitus, Type 2 | Details | |
| Sitagliptin Phosphate/Metformin Hydrochloride | Approved | Merck & Co Inc | Velmetia, Janumet, 捷诺达, Zituvimet | United States | Diabetes Mellitus, Type 2 | Merck & Co Inc | 2007-03-30 | Diabetes Mellitus, Type 2 | Details | |
| Teneligliptin Hydrobromide Hydrate/Canagliflozin Hemihydrate | Approved | Mitsubishi Tanabe Pharma Corp, Daiichi Sankyo Co Ltd | Canalia | Japan | Diabetes Mellitus, Type 2 | Daiichi Sankyo Co Ltd, Mitsubishi Tanabe Pharma | 2017-07-03 | Diabetes Mellitus, Type 2 | Details | |
| Anagliptin/Metformin Hydrochloride | SK-1501 | Approved | Sanwa Kagaku Kenkyusho Co Ltd, Suzuken | Metoana | Japan | Diabetes Mellitus, Type 2 | Sanwa Kagaku Kenkyusho Co Ltd | 2018-09-21 | Diabetes Mellitus, Type 2 | Details |
| Vildagliptin/Metformin Hydrochloride | LMF-237 | Approved | Novartis Pharma Ag | 宜合瑞, Eucreas, EquMet, Zomarist, Icandra | EU | Diabetes Mellitus, Type 2 | Novartis Europharm Ltd | 2007-11-14 | Diabetes Mellitus, Type 2 | Details |
| Alogliptin Benzoate/Metformin Hydrochloride | CT-L01; CT-L-01 | Approved | Takeda Pharmaceutical Co Ltd | Vipdomet, Inisync, Kazano | United States | Diabetes Mellitus, Type 2 | Takeda Pharmaceuticals U.S.A. Inc | 2013-01-25 | Diabetes Mellitus, Type 2 | Details |
| Anagliptin | SK-0403; CWP-0403 | Approved | Sanwa Kagaku Kenkyusho Co Ltd | Suiny, Beskoa | Japan | Diabetes Mellitus, Type 2 | null | 2012-09-28 | Diabetes Mellitus, Type 2 | Details |
| Dapagliflozin Propanediol Monohydrate/Saxagliptin Hydrochloride | Approved | Astrazeneca Plc | Qtern | EU | Diabetes Mellitus, Type 2 | Astrazeneca Ab | 2016-07-15 | Diabetes Mellitus, Type 2 | Details | |
| Alogliptin Benzoate/Pioglitazone Hydrochloride | SYR-322-4833 BL | Approved | Takeda Pharmaceutical Co Ltd | Nesinaact, Oseni, Liovel, Incresync | Japan | Diabetes Mellitus, Type 2 | Takeda | 2011-07-01 | Diabetes Mellitus, Type 2 | Details |
| Sitagliptin Phosphate Monohydrate | ONO-5435A; ONO-5435; MK-0431; MK-431; L-000224715 | Approved | Merck Sharp & Dohme Corp | Ristaben, Xelevia, Tesavel, Glactiv, 捷诺维, Januvia, DIABIT-IS X | United States | Diabetes Mellitus, Type 2 | Merck & Co Inc | 2006-10-16 | Gastroparesis; Carcinoma, Hepatocellular; Hyperlipidemias; Kidney Failure, Chronic; Diabetes Mellitus; Inflammation; Lymphoma, Non-Hodgkin; Leukemia, Myeloid, Acute; Renal Insufficiency, Chronic; Cardiovascular Diseases; Hepatic Insufficiency; Hepatitis C; Psoriasis; Precursor Cell Lymphoblastic Leukemia-Lymphoma; Cystic Fibrosis; Insulin Resistance; Coronavirus Disease 2019 (COVID-19); Myelodysplastic Syndromes; Graft vs Host Disease; Glucose Intolerance; Atherosclerosis; Metabolic Dysfunction-Associated Steatotic Liver Disease; Diabetes Mellitus, Type 2; Heart Failure; HIV Infections; Prediabetic State; Diabetes Mellitus, Type 1; Leukemia, Myelogenous, Chronic | Details |
| Trelagliptin Succinate | TAK-472; SYR-472; SYR-111472; SYR-111472 succinate | Approved | Takeda Pharmaceutical Co Ltd | Zafatek | Japan | Diabetes Mellitus, Type 2 | Takeda | 2015-03-26 | Diabetes Mellitus, Type 2; Diabetes Mellitus | Details |
| Evogliptin Tartrate/Metformin Hydrochloride | DA-1229_01 | Approved | Dong-A Pharmaceutical Co Ltd | Sugamet XR | South Korea | Diabetes Mellitus, Type 2 | Dong-A Pharmaceutical Co Ltd | 2015-12-01 | Diabetes Mellitus, Type 2 | Details |
| Sitagliptin/Ertugliflozin | Approved | Pfizer Inc | Steglujan | United States | Diabetes Mellitus, Type 2 | Merck & Co Inc | 2017-12-19 | Diabetes Mellitus, Type 2 | Details | |
| Omarigliptin | MK-3102 | Approved | Merck & Co Inc | Marizev | Japan | Diabetes Mellitus, Type 2 | Merck Sharp & Dohme Corp | 2015-09-28 | Diabetes Mellitus, Type 2; Renal Insufficiency, Chronic; Diabetes Mellitus | Details |
| Teneligliptin/Pioglitazone | Approved | Glenmark Pharmaceuticals Ltd | Zita Plus Pio | India | Diabetes Mellitus, Type 2 | Glenmark Pharmaceuticals Ltd | 2022-04-25 | Diabetes Mellitus, Type 2 | Details | |
| Metformin Hydrochloride/Sitagliptin Hydrochloride | Approved | Sandoz Bv | EU | Sandoz Bv | 2019-12-10 | Diabetes Mellitus, Type 2 | Details | |||
| Sitagliptin Tartrate | Approved | Heumann Pharma GmbH & Co Generica KG | Sitagliptin Heumann 25 mg Filmtabletten | EU | Heumann Pharma GmbH & Co Generica KG | 2020-12-14 | Diabetes Mellitus, Type 1; Parkinson Disease; Diabetic Nephropathies | Details | ||
| Sitagliptin Hydrochloride | Approved | Laboratorios Alter Sa | Spain | Diabetes Mellitus, Type 2 | Laboratorios Alter Sa | 2019-03-06 | Diabetes Mellitus, Type 2; Polycystic Ovary Syndrome | Details | ||
| Gemigliptin | Approved | Details | ||||||||
| Linagliptin/Metformin Hydrochloride | GC2129A; GC-2129-A; DW6013 | Approved | Boehringer Ingelheim Gmbh | Jentadueto Xr, Trajenta Duo, Jentadueto, 欧双宁 | United States | Diabetes Mellitus, Type 2 | Boehringer Ingelheim Gmbh | 2012-01-30 | Diabetes Mellitus, Type 2 | Details |
| Linagliptin/Empagliflozin | BI 1356/BI 10773; BI 10773/linagliptin; BI 1356/empagliflozin; Empagliflozin/BI 1356; Linagliptin/BI 10773; BI 10773/BI 1356 | Approved | C.H. Boehringer Sohn Ag & Co. Kg | Tradiance, Glyxambi, Esgliteo | United States | Diabetes Mellitus, Type 2 | Boehringer Ingelheim Gmbh | 2015-01-30 | Diabetes Mellitus, Type 2; Cardiovascular Diseases | Details |
| Vildagliptin | LAF-237; DSP-7238; LAF-237A; NVP-LAF-237 | Approved | Novartis Pharma Ag | 佳维乐, Equa, Jalra, Galvus, Gliptus, Xiliarx | EU | Diabetes Mellitus, Type 2 | Novartis Europharm Ltd | 2007-09-25 | Prediabetic State; Diabetes Mellitus, Type 1; Diabetes Mellitus, Type 2; Renal Insufficiency; Schizophrenia; Kidney Diseases; Insulin Resistance; Dyslipidemias; Diabetes, Gestational; Diabetes Mellitus | Details |
| Saxagliptin Hydrochloride/Metformin Hydrochloride | Approved | Astrazeneca Plc | 安立格, Kombiglyze Xr, Komboglyze | EU | Diabetes Mellitus, Type 2 | Astrazeneca Ab | 2010-11-05 | Diabetes Mellitus, Type 2 | Details | |
| Gemigliptin L-tartrate Sesquihydrate | LC-150444; LC-15-0444 | Approved | Lg Life Sciences Ltd | Gemiglo, Zemiglo | South Korea | Diabetes Mellitus, Type 2 | Lg Life Sciences Ltd | 2012-06-27 | Diabetes Mellitus, Type 2; Hepatic Insufficiency | Details |
| Gemigliptin L-tartrate Sesquihydrate/Metformin Hydrochloride | Approved | Lg Life Sciences Ltd | ZemiMet SR | South Korea | Diabetes Mellitus, Type 2 | Lg Life Sciences Ltd | 2015-01-01 | Diabetes Mellitus, Type 2 | Details | |
| Teneligliptin Hydrobromide Hydrate/Metformin Hydrochloride | Approved | Handok Inc | Tenelia M SR | South Korea | Diabetes Mellitus, Type 2 | Handok Inc | 2015-11-03 | Diabetes Mellitus, Type 2 | Details | |
| Evogliptin | DA-1229 | Approved | Dong-A Pharmaceutical Co Ltd | Suganon, Sugarnon | South Korea | Diabetes Mellitus, Type 2 | Dong-A Pharmaceutical Co Ltd | 2015-10-02 | Diabetes Mellitus, Type 2; Kidney Diseases; Aortic valve diseases; Liver Diseases | Details |
| Saxagliptin Hydrate | BMS-477118; BMS-477118-11; OPC-262 | Approved | Bristol-Myers Squibb Company | 安立泽, Onglyza | EU | Diabetes Mellitus, Type 2 | Astrazeneca Ab | 2009-07-31 | Diabetes Mellitus, Type 1; Diabetes Mellitus, Type 2; Hypoglycemia; Diabetes Mellitus | Details |
| Teneligliptin Hydrobromide Hydrate | MP-513 | Approved | Mitsubishi Tanabe Pharma | Tenelia, Tenglyn | Japan | Diabetes Mellitus, Type 2 | Mitsubishi Tanabe Pharma, Daiichi Sankyo Co Ltd | 2012-06-29 | Diabetes Mellitus, Type 2 | Details |
| Retagliptin Phosphate | SP-2086 Phosphate | Approved | Jiangsu Hengrui Medicine Co Ltd | 瑞泽唐 | Mainland China | Diabetes Mellitus, Type 2 | Jiangsu Hengrui Medicine Co Ltd | 2023-06-27 | Diabetes Mellitus, Type 2; Renal Insufficiency; Diabetes Mellitus | Details |
| Sitagliptin Phosphate Monohydrate/Ipragliflozin L-Proline | MK-0431J | Approved | Merck & Co Inc, Astellas Pharma Inc | Sujanu | Japan | Diabetes Mellitus, Type 2 | null | 2018-03-23 | Diabetes Mellitus, Type 2 | Details |
| Linagliptin | BI-1356; BI-1356-BS | Approved | Boehringer Ingelheim Gmbh | 欧唐宁, Ondero, Trajenta, Trayenta, Trazenta, Tradjenta | United States | Diabetes Mellitus, Type 2 | Boehringer Ingelheim Gmbh | 2011-05-02 | Prediabetic State; Diabetes Mellitus, Type 2; Albuminuria; Renal Insufficiency; Schizophrenia; Respiratory Tract Diseases; Coronavirus Disease 2019 (COVID-19); Hypertension; Endocrine System Diseases; Coronavirus Infections; Metabolic Diseases; Hepatic Insufficiency; Glucose Metabolism Disorders; Inflammation; Diabetes Mellitus; Ventricular Dysfunction, Left | Details |
| Rosuvastatin Calcium/Gemigliptin L-tartrate Sesquihydrate | Approved | Lg Life Sciences Ltd | ZemiStatin | Diabetes Mellitus, Type 2; Dyslipidemias | Details | |||||
| Alogliptin Benzoate | SYR-322; TAK-322 | Approved | Takeda | Nesina, 尼欣那, Vipidia | Japan | Diabetes Mellitus, Type 2 | Takeda | 2010-04-06 | Diabetes Mellitus, Type 2; Acute Coronary Syndrome; Diabetes Mellitus | Details |
| Empagliflozin/Linagliptin/Metformin | Approved | Boehringer Ingelheim Gmbh, Eli Lilly And Company | Trijardy XR | United States | Diabetes Mellitus, Type 2 | Boehringer Ingelheim Gmbh | 2020-01-27 | Diabetes Mellitus, Type 2 | Details | |
| Sitagliptin Phosphate/Metformin Hydrochloride | Approved | Merck & Co Inc | Velmetia, Janumet, 捷诺达, Zituvimet | United States | Diabetes Mellitus, Type 2 | Merck & Co Inc | 2007-03-30 | Diabetes Mellitus, Type 2 | Details | |
| Teneligliptin Hydrobromide Hydrate/Canagliflozin Hemihydrate | Approved | Mitsubishi Tanabe Pharma Corp, Daiichi Sankyo Co Ltd | Canalia | Japan | Diabetes Mellitus, Type 2 | Daiichi Sankyo Co Ltd, Mitsubishi Tanabe Pharma | 2017-07-03 | Diabetes Mellitus, Type 2 | Details | |
| Anagliptin/Metformin Hydrochloride | SK-1501 | Approved | Sanwa Kagaku Kenkyusho Co Ltd, Suzuken | Metoana | Japan | Diabetes Mellitus, Type 2 | Sanwa Kagaku Kenkyusho Co Ltd | 2018-09-21 | Diabetes Mellitus, Type 2 | Details |
| Vildagliptin/Metformin Hydrochloride | LMF-237 | Approved | Novartis Pharma Ag | 宜合瑞, Eucreas, EquMet, Zomarist, Icandra | EU | Diabetes Mellitus, Type 2 | Novartis Europharm Ltd | 2007-11-14 | Diabetes Mellitus, Type 2 | Details |
| Alogliptin Benzoate/Metformin Hydrochloride | CT-L01; CT-L-01 | Approved | Takeda Pharmaceutical Co Ltd | Vipdomet, Inisync, Kazano | United States | Diabetes Mellitus, Type 2 | Takeda Pharmaceuticals U.S.A. Inc | 2013-01-25 | Diabetes Mellitus, Type 2 | Details |
| Anagliptin | SK-0403; CWP-0403 | Approved | Sanwa Kagaku Kenkyusho Co Ltd | Suiny, Beskoa | Japan | Diabetes Mellitus, Type 2 | null | 2012-09-28 | Diabetes Mellitus, Type 2 | Details |
| Dapagliflozin Propanediol Monohydrate/Saxagliptin Hydrochloride | Approved | Astrazeneca Plc | Qtern | EU | Diabetes Mellitus, Type 2 | Astrazeneca Ab | 2016-07-15 | Diabetes Mellitus, Type 2 | Details | |
| Alogliptin Benzoate/Pioglitazone Hydrochloride | SYR-322-4833 BL | Approved | Takeda Pharmaceutical Co Ltd | Nesinaact, Oseni, Liovel, Incresync | Japan | Diabetes Mellitus, Type 2 | Takeda | 2011-07-01 | Diabetes Mellitus, Type 2 | Details |
| Sitagliptin Phosphate Monohydrate | ONO-5435A; ONO-5435; MK-0431; MK-431; L-000224715 | Approved | Merck Sharp & Dohme Corp | Ristaben, Xelevia, Tesavel, Glactiv, 捷诺维, Januvia, DIABIT-IS X | United States | Diabetes Mellitus, Type 2 | Merck & Co Inc | 2006-10-16 | Gastroparesis; Carcinoma, Hepatocellular; Hyperlipidemias; Kidney Failure, Chronic; Diabetes Mellitus; Inflammation; Lymphoma, Non-Hodgkin; Leukemia, Myeloid, Acute; Renal Insufficiency, Chronic; Cardiovascular Diseases; Hepatic Insufficiency; Hepatitis C; Psoriasis; Precursor Cell Lymphoblastic Leukemia-Lymphoma; Cystic Fibrosis; Insulin Resistance; Coronavirus Disease 2019 (COVID-19); Myelodysplastic Syndromes; Graft vs Host Disease; Glucose Intolerance; Atherosclerosis; Metabolic Dysfunction-Associated Steatotic Liver Disease; Diabetes Mellitus, Type 2; Heart Failure; HIV Infections; Prediabetic State; Diabetes Mellitus, Type 1; Leukemia, Myelogenous, Chronic | Details |
| Trelagliptin Succinate | TAK-472; SYR-472; SYR-111472; SYR-111472 succinate | Approved | Takeda Pharmaceutical Co Ltd | Zafatek | Japan | Diabetes Mellitus, Type 2 | Takeda | 2015-03-26 | Diabetes Mellitus, Type 2; Diabetes Mellitus | Details |
| Evogliptin Tartrate/Metformin Hydrochloride | DA-1229_01 | Approved | Dong-A Pharmaceutical Co Ltd | Sugamet XR | South Korea | Diabetes Mellitus, Type 2 | Dong-A Pharmaceutical Co Ltd | 2015-12-01 | Diabetes Mellitus, Type 2 | Details |
| Sitagliptin/Ertugliflozin | Approved | Pfizer Inc | Steglujan | United States | Diabetes Mellitus, Type 2 | Merck & Co Inc | 2017-12-19 | Diabetes Mellitus, Type 2 | Details | |
| Omarigliptin | MK-3102 | Approved | Merck & Co Inc | Marizev | Japan | Diabetes Mellitus, Type 2 | Merck Sharp & Dohme Corp | 2015-09-28 | Diabetes Mellitus, Type 2; Renal Insufficiency, Chronic; Diabetes Mellitus | Details |
| Teneligliptin/Pioglitazone | Approved | Glenmark Pharmaceuticals Ltd | Zita Plus Pio | India | Diabetes Mellitus, Type 2 | Glenmark Pharmaceuticals Ltd | 2022-04-25 | Diabetes Mellitus, Type 2 | Details | |
| Metformin Hydrochloride/Sitagliptin Hydrochloride | Approved | Sandoz Bv | EU | Sandoz Bv | 2019-12-10 | Diabetes Mellitus, Type 2 | Details | |||
| Sitagliptin Tartrate | Approved | Heumann Pharma GmbH & Co Generica KG | Sitagliptin Heumann 25 mg Filmtabletten | EU | Heumann Pharma GmbH & Co Generica KG | 2020-12-14 | Diabetes Mellitus, Type 1; Parkinson Disease; Diabetic Nephropathies | Details | ||
| Sitagliptin Hydrochloride | Approved | Laboratorios Alter Sa | Spain | Diabetes Mellitus, Type 2 | Laboratorios Alter Sa | 2019-03-06 | Diabetes Mellitus, Type 2; Polycystic Ovary Syndrome | Details | ||
| Gemigliptin | Approved | Details | ||||||||
| Linagliptin/Metformin Hydrochloride | GC2129A; GC-2129-A; DW6013 | Approved | Boehringer Ingelheim Gmbh | Jentadueto Xr, Trajenta Duo, Jentadueto, 欧双宁 | United States | Diabetes Mellitus, Type 2 | Boehringer Ingelheim Gmbh | 2012-01-30 | Diabetes Mellitus, Type 2 | Details |
| Linagliptin/Empagliflozin | BI 1356/BI 10773; BI 10773/linagliptin; BI 1356/empagliflozin; Empagliflozin/BI 1356; Linagliptin/BI 10773; BI 10773/BI 1356 | Approved | C.H. Boehringer Sohn Ag & Co. Kg | Tradiance, Glyxambi, Esgliteo | United States | Diabetes Mellitus, Type 2 | Boehringer Ingelheim Gmbh | 2015-01-30 | Diabetes Mellitus, Type 2; Cardiovascular Diseases | Details |
| Vildagliptin | LAF-237; DSP-7238; LAF-237A; NVP-LAF-237 | Approved | Novartis Pharma Ag | 佳维乐, Equa, Jalra, Galvus, Gliptus, Xiliarx | EU | Diabetes Mellitus, Type 2 | Novartis Europharm Ltd | 2007-09-25 | Prediabetic State; Diabetes Mellitus, Type 1; Diabetes Mellitus, Type 2; Renal Insufficiency; Schizophrenia; Kidney Diseases; Insulin Resistance; Dyslipidemias; Diabetes, Gestational; Diabetes Mellitus | Details |
| Saxagliptin Hydrochloride/Metformin Hydrochloride | Approved | Astrazeneca Plc | 安立格, Kombiglyze Xr, Komboglyze | EU | Diabetes Mellitus, Type 2 | Astrazeneca Ab | 2010-11-05 | Diabetes Mellitus, Type 2 | Details |
| Name | Research Code | Research Phase | Company | Indications | Clinical Trials |
|---|---|---|---|---|---|
| Dutogliptin Tartrate | REC-01; PHX-1149; PHX-1149T | Phase 3 Clinical | Phenomix | ST Elevation Myocardial Infarction; Myocardial Infarction; Diabetes Mellitus, Type 2; Kidney Diseases; Leukemia, Myeloid, Acute | Details |
| Yogliptin | Phase 3 Clinical | Sichuan Sunheal Pharmaceutical Co Ltd, Chengdu Easton Biopharmaceuticals Co Ltd | Diabetes Mellitus, Type 2 | Details | |
| CPL2009-0031 | Phase 3 Clinical | Cadila Healthcare Ltd | Diabetes Mellitus, Type 2 | Details | |
| Dapagliflozin formate/Linagliptin | IN-C009; AJU-A51 | Phase 3 Clinical | HK inno.N Corporation, Aju Pharm Co Ltd | Diabetes Mellitus, Type 2 | Details |
| DA5221-B2 | DA5221-B2 | Phase 3 Clinical | Dong-A ST Co Ltd | Diabetes Mellitus, Type 2 | Details |
| LID104 | LID104; LID-104 | Phase 3 Clinical | Ems | Diabetes Mellitus, Type 2 | Details |
| TOTUM•63 | TOTUM-63 | Phase 3 Clinical | Valbiotis | Diabetes Mellitus, Type 2 | Details |
| Dapagliflozin/Sitagliptin | SID-1903; DW-6012 | Phase 3 Clinical | Astrazeneca Sa | Myocardial Infarction; ST Elevation Myocardial Infarction; Diabetes Mellitus, Type 2 | Details |
| Talabostat | PT-100; BXCL-701 | Phase 2 Clinical | Point Therapeutics | Neuroblastoma; Adenocarcinoma; Melanoma; Carcinoma, Non-Small-Cell Lung; Neoplasm Metastasis; Leukemia, Lymphocytic, Chronic, B-Cell; Neoplasms, Germ Cell and Embryonal; Carcinoma, Small Cell; Carcinoma, Pancreatic Ductal; Lung Neoplasms; Leukemia, Myeloid, Acute; Prostatic Neoplasms; Liver Neoplasms; Brain Neoplasms; Sarcoma; Neuroendocrine Tumors; Central Nervous System Neoplasms; Small Cell Lung Carcinoma; Pancreatic Neoplasms; Skin Neoplasms; Myelodysplastic Syndromes; Kidney Neoplasms; Solid tumours; Ovarian Neoplasms | Details |
| TQ-F3083 | TQ-F3083; CT-383 | Phase 2 Clinical | Beijing Centaurus Biopharma Co Ltd, Jiangsu Chia Tai-Tianqing Pharmaceutical Co Ltd | Diabetes Mellitus, Type 2; Diabetes Mellitus | Details |
| Begelomab | SAND-26; BT 5/9 | Phase 2 Clinical | Adienne | Dermatomyositis; Graft vs Host Disease | Details |
| YS-110 | YSCMA; YS-110 | Phase 2 Clinical | Y'S Therapeutics Inc | Mesothelioma | Details |
| Singletine | DC-291407; DC-407; DC291407 | Phase 2 Clinical | Cisen Pharmaceutical Co Ltd, Shanghai Institute Of Materia Medica, Chinese Academy Of Sciences | Diabetes Mellitus, Type 2; Fatty Liver | Details |
| Metformin/Dapagliflozin/Sitagliptin | GMRx4 | Phase 2 Clinical | George Medicines PTY Ltd, George Medicines UK Ltd | Diabetes Mellitus, Type 2 | Details |
| Bogliptin maleate | HL012MA | Phase 2 Clinical | Shanghai Institute Of Materia Medica, Chinese Academy Of Sciences, East China University Of Science And Technology, Shandong Baiji Dichang Pharmaceutical Co Ltd | Diabetes Mellitus, Type 2 | Details |
| Augliptin Hydrochloride | Phase 1 Clinical | Shanghai Sun-Sail Pharmaceutical Science & Technology Co Ltd, Nanjing Changao Pharmaceutical Science And Technologyco Ltd | Diabetes Mellitus, Type 2 | Details | |
| Evogliptin Tartrate | LY-05007 | Phase 1 Clinical | Dong-A Pharmaceutical Co Ltd | Diabetes Mellitus, Type 2 | Details |
| TQ-05510 | TQ-05510 | Phase 1 Clinical | Jiangsu Chia Tai-Tianqing Pharmaceutical Co Ltd, Beijing Centaurus Biopharma Co Ltd | Diabetes Mellitus, Type 2 | Details |
| Lobeglitazone/Sitagliptin | CKD-396 | Phase 1 Clinical | Chong Kun Dang Pharmaceutical Corp | Diabetes Mellitus, Type 2 | Details |
| Sitagliptin/Telmisartan | HDDO-16091 + HDDO-16092; HOB-077; HDDO-1609 | Phase 1 Clinical | Hyundai Pharmaceutical Co Ltd | Hypertension; Diabetes Mellitus | Details |
| TSL-0319 | TSL-0319 | Phase 1 Clinical | Jiangsu Tasly Diyi Pharmaceutical Co Ltd | Diabetes Mellitus, Type 2 | Details |
| Linagliptin/Fimasartan Potassium Trihydrate | Phase 1 Clinical | Boryung Pharmaceutical Co Ltd | Hypertension; Diabetes Mellitus | Details | |
| Retagliptin Phosphate/Metformin Hydrochloride | HRX-0701 | Phase 1 Clinical | Jiangsu Hengrui Medicine Co Ltd | Diabetes Mellitus, Type 2 | Details |
| CGT-2201 | CGT-2201 | Phase 1 Clinical | Cgenetech(Suzhou China) Co Ltd | Diabetes Mellitus, Type 2; Diabetes Mellitus | Details |
| Empagliflozin/Metformin/Sitagliptin (Chong Kun Dang Pharmaceutical) | CKD-379 | Phase 1 Clinical | Chong Kun Dang Pharmaceutical Corp | Diabetes Mellitus, Type 2 | Details |
| Empagliflozin/Metformin/Sitagliptin (Overseas Pharmaceuticals) | Phase 1 Clinical | Taizhou Overseas Pharmaceuticals Co Ltd | Diabetes Mellitus, Type 2 | Details | |
| THDBH-100 | THDBH100; THDBH101 | Phase 1 Clinical | Wuxi Apptec(Shanghai) Co Ltd | Diabetes Mellitus, Type 2 | Details |
| WXSHC-071 | WXSHC-071 | Phase 1 Clinical | Dongbao Zixing (Hangzhou) Biological Medicine Co Ltd | Diabetes Mellitus, Type 2 | Details |
| REC-04 | Clinical | Recardio Inc | Cardiovascular Diseases | Details | |
| Dutogliptin Tartrate/Metformin Hydrochloride | Phenomix Corporation | Details | |||
| Dutogliptin Tartrate | REC-01; PHX-1149; PHX-1149T | Phase 3 Clinical | Phenomix | ST Elevation Myocardial Infarction; Myocardial Infarction; Diabetes Mellitus, Type 2; Kidney Diseases; Leukemia, Myeloid, Acute | Details |
| Yogliptin | Phase 3 Clinical | Sichuan Sunheal Pharmaceutical Co Ltd, Chengdu Easton Biopharmaceuticals Co Ltd | Diabetes Mellitus, Type 2 | Details | |
| CPL2009-0031 | Phase 3 Clinical | Cadila Healthcare Ltd | Diabetes Mellitus, Type 2 | Details | |
| Dapagliflozin formate/Linagliptin | IN-C009; AJU-A51 | Phase 3 Clinical | HK inno.N Corporation, Aju Pharm Co Ltd | Diabetes Mellitus, Type 2 | Details |
| DA5221-B2 | DA5221-B2 | Phase 3 Clinical | Dong-A ST Co Ltd | Diabetes Mellitus, Type 2 | Details |
| LID104 | LID104; LID-104 | Phase 3 Clinical | Ems | Diabetes Mellitus, Type 2 | Details |
| TOTUM•63 | TOTUM-63 | Phase 3 Clinical | Valbiotis | Diabetes Mellitus, Type 2 | Details |
| Dapagliflozin/Sitagliptin | SID-1903; DW-6012 | Phase 3 Clinical | Astrazeneca Sa | Myocardial Infarction; ST Elevation Myocardial Infarction; Diabetes Mellitus, Type 2 | Details |
| Talabostat | PT-100; BXCL-701 | Phase 2 Clinical | Point Therapeutics | Neuroblastoma; Adenocarcinoma; Melanoma; Carcinoma, Non-Small-Cell Lung; Neoplasm Metastasis; Leukemia, Lymphocytic, Chronic, B-Cell; Neoplasms, Germ Cell and Embryonal; Carcinoma, Small Cell; Carcinoma, Pancreatic Ductal; Lung Neoplasms; Leukemia, Myeloid, Acute; Prostatic Neoplasms; Liver Neoplasms; Brain Neoplasms; Sarcoma; Neuroendocrine Tumors; Central Nervous System Neoplasms; Small Cell Lung Carcinoma; Pancreatic Neoplasms; Skin Neoplasms; Myelodysplastic Syndromes; Kidney Neoplasms; Solid tumours; Ovarian Neoplasms | Details |
| TQ-F3083 | TQ-F3083; CT-383 | Phase 2 Clinical | Beijing Centaurus Biopharma Co Ltd, Jiangsu Chia Tai-Tianqing Pharmaceutical Co Ltd | Diabetes Mellitus, Type 2; Diabetes Mellitus | Details |
| Begelomab | SAND-26; BT 5/9 | Phase 2 Clinical | Adienne | Dermatomyositis; Graft vs Host Disease | Details |
| YS-110 | YSCMA; YS-110 | Phase 2 Clinical | Y'S Therapeutics Inc | Mesothelioma | Details |
| Singletine | DC-291407; DC-407; DC291407 | Phase 2 Clinical | Cisen Pharmaceutical Co Ltd, Shanghai Institute Of Materia Medica, Chinese Academy Of Sciences | Diabetes Mellitus, Type 2; Fatty Liver | Details |
| Metformin/Dapagliflozin/Sitagliptin | GMRx4 | Phase 2 Clinical | George Medicines PTY Ltd, George Medicines UK Ltd | Diabetes Mellitus, Type 2 | Details |
| Bogliptin maleate | HL012MA | Phase 2 Clinical | Shanghai Institute Of Materia Medica, Chinese Academy Of Sciences, East China University Of Science And Technology, Shandong Baiji Dichang Pharmaceutical Co Ltd | Diabetes Mellitus, Type 2 | Details |
| Augliptin Hydrochloride | Phase 1 Clinical | Shanghai Sun-Sail Pharmaceutical Science & Technology Co Ltd, Nanjing Changao Pharmaceutical Science And Technologyco Ltd | Diabetes Mellitus, Type 2 | Details | |
| Evogliptin Tartrate | LY-05007 | Phase 1 Clinical | Dong-A Pharmaceutical Co Ltd | Diabetes Mellitus, Type 2 | Details |
| TQ-05510 | TQ-05510 | Phase 1 Clinical | Jiangsu Chia Tai-Tianqing Pharmaceutical Co Ltd, Beijing Centaurus Biopharma Co Ltd | Diabetes Mellitus, Type 2 | Details |
| Lobeglitazone/Sitagliptin | CKD-396 | Phase 1 Clinical | Chong Kun Dang Pharmaceutical Corp | Diabetes Mellitus, Type 2 | Details |
| Sitagliptin/Telmisartan | HDDO-16091 + HDDO-16092; HOB-077; HDDO-1609 | Phase 1 Clinical | Hyundai Pharmaceutical Co Ltd | Hypertension; Diabetes Mellitus | Details |
| TSL-0319 | TSL-0319 | Phase 1 Clinical | Jiangsu Tasly Diyi Pharmaceutical Co Ltd | Diabetes Mellitus, Type 2 | Details |
| Linagliptin/Fimasartan Potassium Trihydrate | Phase 1 Clinical | Boryung Pharmaceutical Co Ltd | Hypertension; Diabetes Mellitus | Details | |
| Retagliptin Phosphate/Metformin Hydrochloride | HRX-0701 | Phase 1 Clinical | Jiangsu Hengrui Medicine Co Ltd | Diabetes Mellitus, Type 2 | Details |
| CGT-2201 | CGT-2201 | Phase 1 Clinical | Cgenetech(Suzhou China) Co Ltd | Diabetes Mellitus, Type 2; Diabetes Mellitus | Details |
| Empagliflozin/Metformin/Sitagliptin (Chong Kun Dang Pharmaceutical) | CKD-379 | Phase 1 Clinical | Chong Kun Dang Pharmaceutical Corp | Diabetes Mellitus, Type 2 | Details |
| Empagliflozin/Metformin/Sitagliptin (Overseas Pharmaceuticals) | Phase 1 Clinical | Taizhou Overseas Pharmaceuticals Co Ltd | Diabetes Mellitus, Type 2 | Details | |
| THDBH-100 | THDBH100; THDBH101 | Phase 1 Clinical | Wuxi Apptec(Shanghai) Co Ltd | Diabetes Mellitus, Type 2 | Details |
| WXSHC-071 | WXSHC-071 | Phase 1 Clinical | Dongbao Zixing (Hangzhou) Biological Medicine Co Ltd | Diabetes Mellitus, Type 2 | Details |
| REC-04 | Clinical | Recardio Inc | Cardiovascular Diseases | Details | |
| Dutogliptin Tartrate/Metformin Hydrochloride | Phenomix Corporation | Details |
This web search service is supported by Google Inc.
