



























 Limited Edition Golden Llama is here! Check out how you can get one.
Limited Edition Golden Llama is here! Check out how you can get one.  Limited Edition Golden Llama is here! Check out how you can get one.
Limited Edition Golden Llama is here! Check out how you can get one.
 Offering SPR-BLI Services - Proteins provided for free!
Offering SPR-BLI Services - Proteins provided for free!  Offering SPR-BLI Services - Proteins provided for free!
Offering SPR-BLI Services - Proteins provided for free!
 Here come GMP Grade Cytokines!Free Sample is available!
Here come GMP Grade Cytokines!Free Sample is available!  Here come GMP Grade Cytokines!Free Sample is available!
Here come GMP Grade Cytokines!Free Sample is available!
> 억제제 스크리닝 키트

In some immune checkpoint pathway, tumor cells can hijack the checkpoint pathways to avoid attacks from the immune system. For example, PD-1 is one of the best characterized checkpoint proteins. The binding between PD-1 and its ligand PD-L1 suppresses T-cell activation and allows cancer cells to escape from body's immune surveillance.
Not only in the immune checkpoint proteins, but also the protein such as PCSK9, which binds to the epidermal growth factor-like repeat A (EGF-A) domain of the low-density lipoprotein receptor (LDLR), inducing LDLR degradation. Inhibition of PCSK9 protein function is currently being explored as a means of lowering cholesterol levels.
Antibody drugs can produce therapeutic effects by blocking or neutralizing the interaction between proteins. In response to this market demand, ACROBiosystems developed ELISA kits that can screen neutralizing antibodies.
| Cat. No. | Product Description | Detection mechanism | Order/Preorder |
|---|
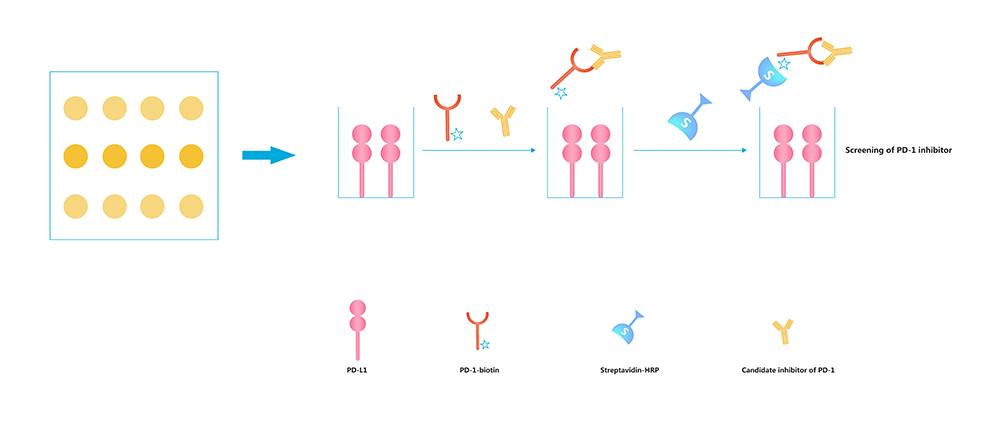
Coat the plate with human PD-L1.
Add Human PD-1-Biotin to bind the coated human PD-L1.
Add your molecule of interest to the tests.
Add Streptavidin-HRP followed by TMB or other colorimetric HRP substrate.
Binding of Biotinylated Human PD-1 to Immobilized Human PD-L1 in a Functional ELISA Assay

Immobilized human PD-L1 protein at 2 μg/mL (100 μL/well) can bind human PD-1-Biotin with a linear range of 0.01 - 0.2μg/mL when detected by Streptavidin-HRP. Background was subtracted from data points before curve fitting.
Inhibition of PD-1-PD-L1 binding by Anti-PD-1 Neutralizing Antibody measured using the PD-1 [Biotinylated]: PD-L1 Inhibitor Screening ELISA Assay Pair (Cat. No. EP-101)
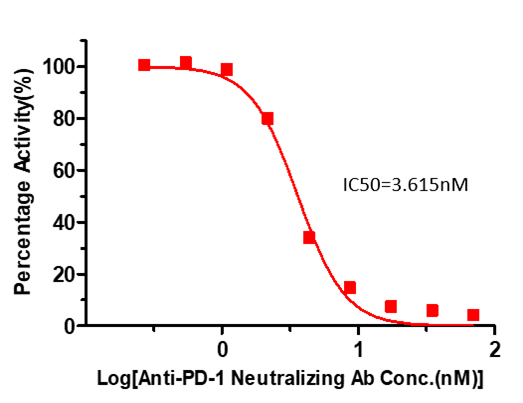
Serial dilutions of anti-PD-1 neutralizing antibody (1:1 serial dilutions, from 10 μg/mL to 0.078 μg/mL (69.628 nM to 0.544 nM) was added into PD-L1 : PD-1-Biotin binding reactions. The assay was performed according to the above described protocol. Background was subtracted from data points prior to log transformation and curve fitting.
This web search service is supported by Google Inc.






