
주문제작 서비스 문의
찾으시는 제품이 저희 사이트에 없으신가요? 단백질 주문 제작 서비스를 이용해보세요!
주문제작 서비스 문의 >>
























 Offering SPR-BLI Services - Proteins provided for free!
Offering SPR-BLI Services - Proteins provided for free!  Offering SPR-BLI Services - Proteins provided for free!
Offering SPR-BLI Services - Proteins provided for free!
 Fill out organ-on-a-chip questionnaire to win a FREE gift!
Fill out organ-on-a-chip questionnaire to win a FREE gift!  Fill out organ-on-a-chip questionnaire to win a FREE gift!
Fill out organ-on-a-chip questionnaire to win a FREE gift!
 Here come GMP Grade Cytokines!Free Sample is available!
Here come GMP Grade Cytokines!Free Sample is available!  Here come GMP Grade Cytokines!Free Sample is available!
Here come GMP Grade Cytokines!Free Sample is available!
| ID | Components | Size |
| EP120-C01 | High-bind Plate | 1 plate |
| EP120-C02 | Human PD-L2 | 50 μg |
| EP120-C03 | Biotinylated Human PD-1 | 10 μg |
| EP120-C04 | Anti-PD-1 Neutralizing Antibody | 20 μg |
| EP120-C05 | Streptavidin-HRP | 10 μg |
| EP120-C06 | Coating Buffer | 12 mL |
| EP120-C07 | 10xWashing Buffer | 50 mL |
| EP120-C08 | Blocking Buffer | 50 mL |
| EP120-C09 | Substrate Solution | 24 mL |
| EP120-C10 | Stop Solution | 7 mL |
This kit is developed for screening for inhibitors of human PD1 binding to human PD-L2.
It is for research use only.
The opened kit should be stored per components table. The shelf life is 30 days from the date of opening.
Your experiment will include 4 simple steps:
a) Coat the plate with human PD-L2.
b) Add your molecule of interest to the tests.
c) Add human PD-1-Biotin to bind the coated human PD-L2.
d) Add Streptavidin-HRP followed by TMB or other colorimetric HRP substrate.
Finally, the half maximal inhibitory concentration (IC50) of your compound to PD-L2: PD-1 binding will be determined by comparing OD readings among different experimental groups.

INHIBITION OF PD-1 [BIOTINYLATED]: PD-L2 BINDING BY ANTI-PD-1 NEUTRALIZING ANTIBODY
Serial dilutions of Anti-PD-1 Neutralizing antibody (Catalog # EP120-C04) (1:1 serial dilution, from 5 μg/mL to 0.01μg/mL) was added into PD-L2: Biotinlated PD-1binding reactions. The assay was performed according to the protocol described above. Background was subtracted from data points prior to log transformation and curve fitting (QC tested).

ACROBiosystems는 생체 활성이 높은 균질한 CD3 δ/CD3 ε 및 CD3 γ/CD3 ε 단백질을 개발하여 이중특이 항체를 치료제로의 임상 개발을 가속시킵니다.

오가노이드 툴 박스는 약물 개발 프로젝트의 진행을 가속할 수 있는 레디메이드 오가노이드와 오가노이드 분화 툴 키트 및 다양한 서비스를 포함하는 오가노이드 솔루션 모음입니다.
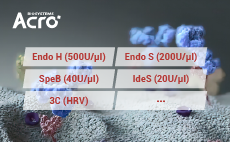
항체 및 번역 후 변형(PTMs)의 특성 분석을 위해, ACROBiosysyems는 IdeS, SpeB, EndoH, and Endo S proteases 계열의 효소를 개발하였습니다.

고품질 GMP 등급 사이토카인 IL-15,IL-7, IL-21 등 제품은 신속한 임상/신약승인신청을 위해 개발된 것입니다.

50+ car-t의 인기 타겟을 커버하고, car 발현 검사를 위해 특별히 설계된 항원 단백질은 스트림 검증을 통해 고특이성 car 발현을 검출할 수 있으며, 고 배치간 일관성을 가지고 있다.pe/fi표기 단백질로 한 단계 염색하면 고특이성, 무 백그라운드로 car 발현 검사를 검사할 수 있습니다.

GMP 등급 사이토카인. 고품질 세포 활성화 및 확장 시약. 유전자 편집 시약, 효소 CAR 검사 시약 등 제품. 세포 및 유전자 요법 고객님에게 포괄적인 솔루션을 제공하고 초기 약물 발견에서 임상 연구에 이르기까지 모든 단계에서 귀하와 동반하겠습니다.

cd20, claudin18.2, cd133, gprc5d, ccr5, ccr8을 포함한 안정적이고 활성이 높은 전장 다중 막관통 단백질은 면역, elisa, spr, bli, 세포 실험, car 양성률 검사 등에 광범위하게 사용된다. vlp, 스케일제거제, nanodisc의 다양한 기술 플랫폼은 다중 막관통 단백질을 목표로 하는 약물 연구에 도움을 준다.

현재 이미 알려진 거의 대부분의 면역 체크 포인트 분자를 포함하여 다양한 레벨과 물종을 제공하여 고객이 선택할수 있도록 하고있다. 천연중합체 형식은 mals를 통해 검증되었고 생물활성은 elisa/spr/bli/facs 등을 통해 검증되었으며 또한 biotin/fitc 표지는 높은 처리량을 가진 항체를 선별하는데 편리하다.

ACROBiosystems는 ADC약물의 연구개발을 돕기 위해 노력하고 있으며, 다양한 인기 타겟에 서로 다른 종, 다른 라벨의 제품을 개발했으며, 높은 순도, 높은 친화력 등 특징을 가지고 있으며, 면역, 항체 선택, spr, 세포 활성 검사 및 기타 실험에 적합하고, 해당 protocol을 무료로 제공합니다.

Fc 수용체 단백질은 모든 분자를 포함하고 있을 뿐만 아니라, 일반적인 돌연변이와 바이오틴 표기 종류을 포함하고 있어 고객들이 단일 클론 항체 개발을 가속화할 수 있다.

포괄적으로 풍부한 사이토카인 표적 단백질은 HEK293 진핵생물 시스템에 의해 발현되며 천연 구조에 더 가깝다. 고순도는 SDS-PAGE/HPLC/MALS에 의해 검증된다. 높은 생물학적 활성이 ELISA/SPR/BLI에 의해 검증된다. 배치간 일관성이 우수하여 항체 면역, 스크리닝 및 품질 관리 과정에 적합하다.

Aneuro는 ACROBiosystems가 뇌과학 연구에 초점을 맞춘 제품 라인으로서 치료 및 진단 연구 단백질, PFFs와 재조합 신경 인자 등 뇌과학 연구 분야에 중요한 단백질 제품을 제공하여 뇌과학 연구에 도움이 된다.
| English Name | Research Code | Research Phase | Company | First Brand Name | First Approved Country | First Indication | First Approved Company | First Approved Date | Indications | Clinical Trials |
|---|---|---|---|---|---|---|---|---|---|---|
| Zimberelimab | AB-122; GLS-010; WBP-3055 | Approved | Wuxi Biologics (Shanghai) Co Ltd, Harbin Gloria Pharmaceuticals Co Ltd | 誉妥, YUTUO | Mainland China | Hodgkin Disease | Guangzhou Gloria Biosciences Co Ltd | 2021-08-25 | Prostatic Neoplasms, Castration-Resistant; Adenocarcinoma; Melanoma; Uterine Cervical Neoplasms; Carcinoma, Non-Small-Cell Lung; Esophageal adenocarcinoma; Endometrial Neoplasms; Carcinoma, Pancreatic Ductal; Lung Neoplasms; Lymphoma, Non-Hodgkin; Carcinoma, Squamous Cell; Colorectal Neoplasms; Breast Neoplasms; Multiple Myeloma; Urinary Bladder Neoplasms; Solid tumours; Neoplasms; Triple Negative Breast Neoplasms; Hodgkin Disease; Glioblastoma; Pancreatic Neoplasms; Lymphoma, Large B-Cell, Diffuse; Squamous Cell Carcinoma of Head and Neck; Esophageal Neoplasms; Carcinoma, Renal Cell; Carcinoma, Merkel Cell; Stomach Neoplasms; Liver Neoplasms; Biliary Tract Neoplasms; Ovarian Neoplasms | Details |
| Penpulimab | AK-105 | Approved | Zhongshan Akeso Biopharma Co Ltd | 安尼可, ANNIKO | Mainland China | Hodgkin Disease | Zhengda Tianqing Kangfang (Shanghai) Biomedical Technology Co Ltd | 2021-08-03 | Neuroendocrine Tumors; Gastrointestinal Neoplasms; Carcinoma, Non-Small-Cell Lung; Carcinoma, Hepatocellular; Lung Neoplasms; Carcinoma, Squamous Cell; Lymphoma, Non-Hodgkin; Urologic Neoplasms; Nasopharyngeal Carcinoma; Solid tumours; Microsatellite instability-high cancer; Neoplasms; Small Cell Lung Carcinoma; Hodgkin Disease; Esophageal Neoplasms; Thoracic Neoplasms; Head and Neck Neoplasms; Liver Neoplasms | Details |
| Toripalimab | TAB-001; JS-001; SO-001; JS-001sc; CHS-007 | Approved | Shanghai Junshi Biosciences Co Ltd, Suzhou Union Biopharm Biosciences Co Ltd | 拓益, LOQTORZI | Mainland China | Melanoma | Shanghai Junshi Biosciences Co Ltd | 2018-12-17 | Lymphoma; Rejection of organ transplantation; Urinary Bladder Neoplasms; Sarcoma; Breast Neoplasms; Nasopharyngeal Carcinoma; Cholangiocarcinoma; Colorectal Neoplasms; Urologic Neoplasms; Neuroendocrine Tumors; Epstein-Barr Virus Infections; Esophageal Squamous Cell Carcinoma; Laryngeal Neoplasms; Lung Neoplasms; Melanoma; Carcinoma, Hepatocellular; Carcinoma, Non-Small-Cell Lung; Adenocarcinoma; Stomach Neoplasms; Solid tumours; Colorectal Neoplasms, Hereditary Nonpolyposis; Head and Neck Neoplasms; Kidney Neoplasms; Carcinoma, Renal Cell; Anus Neoplasms; Esophageal Neoplasms; Squamous Cell Carcinoma of Head and Neck; Biliary Tract Neoplasms; Pain; Triple Negative Breast Neoplasms; Small Cell Lung Carcinoma; Neoplasms; Carcinoma, Transitional Cell; Nasopharyngeal Neoplasms; Nasopharyngeal Diseases; Hypopharyngeal Neoplasms | Details |
| Prolgolimab | BCD-100 | Approved | Biocad | Forteca | Russian Federation | Melanoma | Biocad | 2020-04-01 | Carcinoma, Renal Cell; Lung Neoplasms; Melanoma; Carcinoma, Non-Small-Cell Lung; Uterine Cervical Neoplasms | Details |
| Cadonilimab | AK-104 | Approved | Zhongshan Akeso Biopharma Co Ltd | 开坦尼 | Mainland China | Uterine Cervical Neoplasms | Kangfang Pharmaceutical Co Ltd | 2022-06-29 | Microsatellite Instability; Adenocarcinoma; Carcinoma, Hepatocellular; Uterine Cervical Neoplasms; Carcinoma, Non-Small-Cell Lung; Melanoma; Esophageal Squamous Cell Carcinoma; Colorectal Neoplasms; DNA Repair-Deficiency Disorders; Urologic Neoplasms; Vulvar Neoplasms; Nasopharyngeal Carcinoma; Solid tumours; Microsatellite instability-high cancer; Pancreatic Neoplasms; Small Cell Lung Carcinoma; Neoplasms; Triple Negative Breast Neoplasms; Vaginal Neoplasms; Esophageal Neoplasms; Stomach Neoplasms; Carcinoma, Renal Cell; Carcinoma; Lymphoma, T-Cell, Peripheral | Details |
| Cemiplimab | REGN-2810; SAR-439684 | Approved | Regeneron Pharmaceuticals Inc | Libtayo | United States | Carcinoma, Squamous Cell | Regeneron Pharmaceuticals Inc | 2018-09-28 | Mouth Neoplasms; Sarcoma; Peritoneal Neoplasms; Diffuse Intrinsic Pontine Glioma; Carcinoma, Squamous Cell; Lung Neoplasms; Brain metastases; Fallopian Tube Neoplasms; Carcinoma, Brown-Pearce; Prostatic Neoplasms; Glioma; Endometrial Neoplasms; Carcinoma, Non-Small-Cell Lung; Neoplasm Metastasis; Uterine Cervical Neoplasms; Mycosis Fungoides; Melanoma; Carcinoma, Hepatocellular; Skin Neoplasms; Solid tumours; HIV Infections; Carcinoma, Bronchogenic; Carcinoma, Basal Cell; Squamous Cell Carcinoma of Head and Neck; Carcinoma, Merkel Cell; Hemangiosarcoma; Carcinoma, Renal Cell; Ovarian Neoplasms; Carcinoma, Transitional Cell; Neoplasms; Hodgkin Disease; Colonic Neoplasms; Papillomavirus Infections; Central Nervous System Neoplasms; Multiple Myeloma; Hepatitis B | Details |
| Nivolumab | MDX-1106; BMS-936558; BMS-936558-01; ono-0123; ONO-4538 | Approved | Ono Pharmaceutical Co Ltd, Bristol-Myers Squibb Company | 欧狄沃, Opdivo | Japan | Melanoma | Ono Pharmaceutical Co Ltd | 2014-07-04 | Liver Neoplasms; Hematologic Neoplasms; Solid tumours; Tobacco Smoking; Drug-Related Side Effects and Adverse Reactions; Biliary Tract Neoplasms; Acquired Immunodeficiency Syndrome; Head and Neck Neoplasms; Ovarian Neoplasms; Bone Marrow Neoplasms; Colorectal Neoplasms, Hereditary Nonpolyposis; Lymphoma, T-Cell, Peripheral; Leiomyosarcoma; Kidney Neoplasms; Ependymoma; Medulloblastoma; Leukemia, Myelogenous, Chronic; Hematopoietic stem cell transplantation (HSCT); HIV Infections; Esophageal Neoplasms; Carcinoma, Renal Cell; Squamous Cell Carcinoma of Head and Neck; Stomach Neoplasms; Mismatch Repair Deficient Cancer; Anus Neoplasms; Leukoplakia, Oral; Leukemia, Lymphoid; Carcinoma, Merkel Cell; Polycythemia Vera; Carcinoma; Giant Cell Arteritis; Thoracic Neoplasms; Inflammatory Bowel Diseases; Dermatomyositis; Neoplasms; Nasopharyngeal Neoplasms; Carcinoma, Transitional Cell; Small Cell Lung Carcinoma; Hodgkin Disease; Autoimmune Diseases; Myelodysplastic Syndromes; Pancreatic Neoplasms; Adrenal Cortex Neopla | Details |
| Nivolumab/Relatlimab | BMS-986213; BMS-936558/BMS-986016 | Approved | Bristol-Myers Squibb Company | OPDUALAG | United States | Melanoma | Bristol-Myers Squibb Company | 2022-03-18 | Solid tumours; Esophageal Neoplasms; Stomach Neoplasms; Carcinoma, Merkel Cell; Colorectal Neoplasms; Melanoma | Details |
| Sintilimab | IBI-308 | Approved | Innovent Biologics(Suzhou) Co Ltd | Tyvyt, 达伯舒 | Mainland China | Hodgkin Disease | Innovent Biologics(Suzhou) Co Ltd | 2018-12-24 | Lung Diseases; Adenocarcinoma; Carcinoma, Hepatocellular; Carcinoma, Non-Small-Cell Lung; Lymphoma, Non-Hodgkin; Neoplasms, Unknown Primary; Lung Neoplasms; Esophageal Squamous Cell Carcinoma; Colorectal Neoplasms; Sarcoma; Lymphoma, Extranodal NK-T-Cell; Lymphoma, T-Cell, Peripheral; Nasopharyngeal Neoplasms; Hodgkin Disease; Respiratory Tract Diseases; Neoplasms; Hemangiosarcoma; Thoracic Neoplasms; Esophageal Neoplasms; Stomach Neoplasms; Liver Neoplasms; Solid tumours | Details |
| Serplulimab | HLX-10 | Approved | Shanghai Henlius Biotech Inc | 汉斯状, Zerpidio, Han Si Zhuang, HANSIZHUANG | Mainland China | Microsatellite instability-high cancer | Shanghai Henlius Biopharmaceuticals Co Ltd | 2022-03-22 | Triple Negative Breast Neoplasms; Uterine Cervical Neoplasms; Carcinoma, Hepatocellular; Carcinoma, Non-Small-Cell Lung; Carcinoma, Squamous Cell; Esophageal Squamous Cell Carcinoma; Colorectal Neoplasms; Microsatellite Instability; Microsatellite instability-high cancer; Neoplasms; Solid tumours; Small Cell Lung Carcinoma; Hepatitis B, Chronic; Carcinoma; Squamous Cell Carcinoma of Head and Neck; Esophageal Neoplasms; Stomach Neoplasms; Liver Neoplasms; Head and Neck Neoplasms | Details |
| Tislelizumab | BGB-A317; VDT-482 | Approved | Beigene Ltd | 百泽安, Tevimbra, TEVIMBRA | Mainland China | Hodgkin Disease | Beigene (Shanghai) Biotechnology Co Ltd | 2019-12-26 | Lung Neoplasms; Lymphoma, Extranodal NK-T-Cell; Lymphoma, Mantle-Cell; Cholangiocarcinoma; Lymphoma, Follicular; Colorectal Neoplasms; Esophageal Squamous Cell Carcinoma; Lymphoma, T-Cell, Cutaneous; Lymphoma; Carcinoma, Squamous Cell; Nasopharyngeal Carcinoma; Metastatic breast cancer; Lymphoma, Large-Cell, Anaplastic; Lymphoma, T-Cell; Carcinoma, Hepatocellular; Carcinoma, Non-Small-Cell Lung; Melanoma; Uterine Cervical Neoplasms; Leukemia, Lymphocytic, Chronic, B-Cell; Mismatch Repair Deficient Cancer; Lymphoma, T-Cell, Peripheral; Liver Neoplasms; Lymphoma, B-Cell, Marginal Zone; Leukemia; Stomach Neoplasms; Esophageal Neoplasms; Squamous Cell Carcinoma of Head and Neck; Rectal Neoplasms; Solid tumours; Lymphoma, Large B-Cell, Diffuse; Neoplasms; Hodgkin Disease; Carcinoma, Transitional Cell; Small Cell Lung Carcinoma; Microsatellite instability-high cancer; Urinary Bladder Neoplasms; Digestive System Neoplasms | Details |
| Retifanlimab | MGA-012; INCMGA-0012; INCMGA-00012 | Approved | Macrogenics Inc | ZYNYZ | United States | Carcinoma, Merkel Cell | Incyte Corp | 2023-03-22 | Carcinoma, Squamous Cell; Neuroendocrine Tumors; Liposarcoma; Breast Neoplasms; Lymphoma, Follicular; Sarcoma; Astrocytoma; Colorectal Neoplasms; Carcinoma, Pancreatic Ductal; Glioma; Digestive System Neoplasms; Endometrial Neoplasms; Esophageal adenocarcinoma; Penile Neoplasms; Neoplasm Metastasis; Carcinoma, Endometrioid; Carcinoma, Non-Small-Cell Lung; Carcinoma, Hepatocellular; Gastrointestinal Neoplasms; Melanoma; Neoplasms; Solid tumours; Ovarian Neoplasms; Lymphoma, B-Cell; Squamous Cell Carcinoma of Head and Neck; Carcinoma, Renal Cell; Anus Neoplasms; Esophageal Neoplasms; Stomach Neoplasms; Carcinoma, Merkel Cell; Head and Neck Neoplasms; Pancreatic Neoplasms; Triple Negative Breast Neoplasms; Carcinoma, Transitional Cell; Malignant Carcinoid Syndrome; Lymphoma, Large B-Cell, Diffuse; Glioblastoma; Prostatic Neoplasms, Castration-Resistant; Oligodendroglioma; Urinary Bladder Neoplasms | Details |
| Dostarlimab | WBP-285; TSR-042; ANB-011; GSK-4057190 | Approved | Anaptysbio Inc | JEMPERLI, Jemperli | EU | Endometrial Neoplasms | Glaxosmithkline (Ireland) Ltd | 2021-04-21 | Sarcoma, Clear Cell; Neoplasms, Second Primary; Melanoma; Uterine Cervical Neoplasms; Carcinoma, Non-Small-Cell Lung; Carcinoma, Neuroendocrine; Fallopian Tube Neoplasms; Lung Neoplasms; Endometrial Neoplasms; Peritoneal Neoplasms; Colorectal Neoplasms; Breast Neoplasms; Solid tumours; Gestational Trophoblastic Disease; Mesothelioma; Small Cell Lung Carcinoma; Colonic Neoplasms; Pancreatic Neoplasms; Neoplasms; Rectal Neoplasms; Mismatch Repair Deficient Cancer; Head and Neck Neoplasms; Ovarian Neoplasms | Details |
| Socazolimab | STIA-1014; ZKAB-001; STI-1014; ZKAB001; STI-A1014 | Approved | Zhaoke (Guangzhou) Oncology Pharmaceutical Ltd | Mainland China | Uterine Cervical Neoplasms | Zhaoke (Guangzhou) Oncology Pharmaceutical Ltd | 2023-12-19 | Biliary Tract Neoplasms; Esophageal Neoplasms; Carcinoma, Transitional Cell; Small Cell Lung Carcinoma; Osteosarcoma; Bile Duct Neoplasms; Esophageal Squamous Cell Carcinoma; Melanoma; Uterine Cervical Neoplasms | Details | |
| Pucotenlimab | AK-103; HX-008 | Approved | Taizhou Hanzhong Biomedical Co Ltd, Hanx Biopharmaceutical Co Ltd, Zhongshan Akeso Biopharma Co Ltd, Lepu Biotech Co Ltd | 普佑恒 | Mainland China | Microsatellite instability-high cancer; Mismatch Repair Deficient Cancer | Lepu Biotech Co Ltd | 2022-07-19 | Solid tumours; Stomach Neoplasms; Esophageal Neoplasms; Mismatch Repair Deficient Cancer; Thyroid Carcinoma, Anaplastic; Triple Negative Breast Neoplasms; Urinary Bladder Neoplasms; Microsatellite instability-high cancer; Colorectal Neoplasms; Melanoma; Carcinoma, Hepatocellular; Carcinoma, Non-Small-Cell Lung; Adenocarcinoma; Gastrointestinal Neoplasms | Details |
| Camrelizumab | INCSHR-1210; HR-301210; SHR-1210; SHR01210 | Approved | Jiangsu Hengrui Medicine Co Ltd, Suzhou Suncadia Biopharmaceuticals Co Ltd | AiRuiKa, 艾立妥, 艾瑞卡 | Mainland China | Hodgkin Disease | Suzhou Suncadia Biopharmaceuticals Co Ltd | 2019-05-29 | Lung Neoplasms; Nasopharyngeal Carcinoma; Breast Neoplasms; Lymphoma, Extranodal NK-T-Cell; Cholangiocarcinoma; Sarcoma; Colorectal Neoplasms; Genital Neoplasms, Female; Urologic Neoplasms; Esophageal adenocarcinoma; Esophageal Squamous Cell Carcinoma; Lymphoma; Respiratory Tract Neoplasms; Carcinoma, Squamous Cell; Endometrial Neoplasms; Paranasal Sinus Neoplasms; Uterine Neoplasms; Uterine Cervical Neoplasms; Carcinoma, Hepatocellular; Carcinoma, Non-Small-Cell Lung; Melanoma; Gastrointestinal Neoplasms; Neoplasm Metastasis; Esophageal Diseases; Neoplasms; Biliary Tract Neoplasms; Liver Neoplasms; Carcinoma, Bronchogenic; Ovarian Neoplasms; Hemorrhagic Fever, Ebola; Esophageal Neoplasms; Stomach Neoplasms; Bronchial Neoplasms; Thoracic Neoplasms; Carcinoma, Renal Cell; Rectal Neoplasms; Solid tumours; Triple Negative Breast Neoplasms; Hodgkin Disease; Nasopharyngeal Neoplasms; Small Cell Lung Carcinoma; Hypopharyngeal Neoplasms; Carcinoma, Transitional Cell; Respiratory Tract Diseases; Lymphoma, Large B-Cel | Details |
| Pembrolizumab | SCH-900475; h409A11; MK-3475 | Approved | Merck & Co Inc | 可瑞达, Keytruda | United States | Melanoma | Merck Sharp & Dohme Corp | 2014-09-04 | Solid tumours; Biliary Tract Neoplasms; Lymphoma, B-Cell; HIV Infections; Hematologic Neoplasms; Liver Neoplasms; Ovarian Neoplasms; Head and Neck Neoplasms; Kidney Neoplasms; Ependymoma; Colorectal Neoplasms, Hereditary Nonpolyposis; Medulloblastoma; Bone metastases; Intestinal Neoplasms; Erythema; Ovarian germ cell tumor; Leiomyosarcoma; Lymphoma, T-Cell, Peripheral; Lymphoma, B-Cell, Marginal Zone; Neoplasm, Residual; Neoplasm Recurrence, Local; Thyroid Diseases; Anus Neoplasms; Stomach Neoplasms; Squamous Cell Carcinoma of Head and Neck; Esophageal Neoplasms; Carcinoma, Renal Cell; Polycythemia Vera; Carcinoma; Carcinoma, Merkel Cell; Carcinoid Tumor; Rectal Neoplasms; Vaginal Diseases; Spinal Cord Neoplasms; Mismatch Repair Deficient Cancer; Vaginitis; Pheochromocytoma; Cystadenocarcinoma, Serous; Leukemia, Lymphoid; Genital Neoplasms, Male; Thoracic Neoplasms; Neoplasms; Small Cell Lung Carcinoma; Lymphoma, Large B-Cell, Diffuse; Carcinoma, Transitional Cell; Hodgkin Disease; Salivary Gland Neoplasms; I | Details |
| English Name | Research Code | Research Phase | Company | Indications | Clinical Trials |
|---|---|---|---|---|---|
| PD-1 Monoclonal Antibody(Sixth Affiliated Hospital Sun Yat-Sen University) | Phase 2 Clinical | Sixth Affiliated Hospital Sun Yat-Sen University | Neoplasms; Colorectal Neoplasms | Details | |
| INCA-32459 | INCA-32459 | Phase 1 Clinical | Incyte Corp, Merus Nv | Neoplasms | Details |
| Recombinant humanized anti-PD-1 monoclonal antibody (Shanghai CP Guojian) | 609-A; SSGJ-609A; SSGJ-609-A | Phase 2 Clinical | Solid tumours; Sarcoma; Breast Neoplasms | Details | |
| Peramprizumab | Phase 2 Clinical | Sun Yat-Sen University | Nasopharyngeal Carcinoma | Details | |
| IBI-321 | IBI-321 | Phase 1 Clinical | Innovent Biologics(Suzhou) Co Ltd | Solid tumours; Neoplasms | Details |
| IAP-0971 | IAP-0971 | Phase 2 Clinical | SunHo (China) BioPharmaceutical Co Ltd | Solid tumours; Neoplasms; Urinary Bladder Neoplasms | Details |
| EGFRvIII-directed CAR-T cell tharapy (Novartis/University of Pennsylvania) | LXF-821 | Phase 1 Clinical | University Of Pennsylvania, Novartis Pharma Ag | Glioblastoma | Details |
| Izuralimab | XmAb-23104; XmAb-104 | Phase 2 Clinical | Xencor Inc | Sarcoma; Uterine Cervical Neoplasms; Carcinoma, Hepatocellular; Carcinoma, Non-Small-Cell Lung; Melanoma; Endometrial Neoplasms; Colorectal Neoplasms; Breast Neoplasms; Nasopharyngeal Carcinoma; Solid tumours; Small Cell Lung Carcinoma; Pancreatic Neoplasms; Carcinoma, Transitional Cell; Squamous Cell Carcinoma of Head and Neck; Carcinoma, Renal Cell; Stomach Neoplasms; Esophageal Neoplasms | Details |
| TC-510 | TC-510 | Phase 2 Clinical | Tcr2 Therapeutics Inc | Ovarian Neoplasms; Solid tumours; Pancreatic Neoplasms; Triple Negative Breast Neoplasms; Mesothelioma; Cholangiocarcinoma; Colorectal Neoplasms; Carcinoma, Non-Small-Cell Lung; Adenocarcinoma | Details |
| ONO-4685 | ONO-4685 | Phase 1 Clinical | Merus Nv | Psoriasis; Lymphoma, T-Cell; Plaque psoriasis | Details |
| Rosnilimab | ANB-030 | Phase 2 Clinical | Anaptysbio Inc | Alopecia Areata; Arthritis, Rheumatoid; Colitis, Ulcerative | Details |
| CA-170 | CA-170; AUPM-170 | Phase 1 Clinical | Aurigene | Solid tumours | Details |
| Anti-CTLA-4/PD-1 expressing EGFR-CAR-T | Phase 2 Clinical | Shanghai Cell Therapy Research Institute | Solid tumours | Details | |
| Anti-PD1/anti-TIM3 bispecific antibody (L&L vision biopharmaceuticals) | Bis-5 | Phase 1 Clinical | L&L Biopharma Co Ltd | Neoplasms | Details |
| TY-101 | TY-101; TY101 | Phase 2 Clinical | Tayu Huaxia Biotech Medical Group Co Ltd | Solid tumours; Lymphoma | Details |
| Tobemstomig | RO-7247669 | Phase 2 Clinical | F. Hoffmann-La Roche Ltd | Solid tumours; Liver Neoplasms; Carcinoma; Carcinoma, Renal Cell; Neoplasms; Carcinoma, Transitional Cell; Breast Neoplasms; Esophageal Squamous Cell Carcinoma; Melanoma; Carcinoma, Non-Small-Cell Lung; Carcinoma, Hepatocellular | Details |
| Pradusinstobart | LVGN-3616; SSI-361 | Phase 2 Clinical | Lyvgen Biopharma(HK)Ltd | Head and Neck Neoplasms; Solid tumours; Ovarian Neoplasms; Squamous Cell Carcinoma of Head and Neck; Esophageal Neoplasms; Stomach Neoplasms; Neoplasms; Papillomavirus Infections; Uveal melanoma; Sarcoma; Carcinoma, Hepatocellular; Neoplasm Metastasis | Details |
| BAT-3306 | BAT3306; BAT-3306 | Phase 1 Clinical | Bio-Thera Solutions Ltd | Details | |
| Lomvastomig | RG-7769; RO-7121661 | Phase 2 Clinical | F. Hoffmann-La Roche Ltd | Solid tumours; Neoplasms; Small Cell Lung Carcinoma; Esophageal Squamous Cell Carcinoma; Carcinoma, Non-Small-Cell Lung; Melanoma | Details |
| SSGJ-706 | SSGJ-706 | Phase 1 Clinical | Solid tumours; Lymphoma | Details | |
| SSGJ-705 | SSGJ-705 | Phase 1 Clinical | Solid tumours; Neoplasms | Details | |
| PD-1 Inhibitor Therapy(Sun Yat-Sen Memorial Hospital of Sun Yat-Sen University) | Phase 3 Clinical | Sun Yat-Sen Memorial Hospital Of Sun Yat-Sen University | Rejection of liver transplantation; Squamous Cell Carcinoma of Head and Neck; Carcinoma, Hepatocellular | Details | |
| RG-6279 | RG-6279 | Phase 1 Clinical | F. Hoffmann-La Roche Ltd | Solid tumours | Details |
| JS-201 | JS-201 | Phase 1 Clinical | Shanghai Junshi Biosciences Co Ltd | Neoplasms | Details |
| ASKG-915 | ASKG-915 | Phase 1 Clinical | Askgene Pharma | Solid tumours; Neoplasms | Details |
| GME-751 | GME751; GME-751 | Phase 3 Clinical | Sandoz | Melanoma; Carcinoma, Non-Small-Cell Lung | Details |
| LBL-015 | LBL-015 | Phase 2 Clinical | Nanjing Leads Biolabs Co Ltd | Solid tumours | Details |
| Anti-CTLA-4 and PD-1 CAR-T cell therapy (Shanghai Cell Therapy Research Institute) | Phase 2 Clinical | Shanghai Cell Therapy Research Institute | Neoplasms | Details | |
| Pimivalimab | JTX-4014 | Phase 2 Clinical | Jounce Therapeutics Inc | Neoplasms; Carcinoma, Non-Small-Cell Lung | Details |
| HX-009 | HX-009 | Phase 2 Clinical | Hanx Biopharmaceutical Co Ltd, Wuhan Hanxiong Biotechnology Co Ltd | Solid tumours; Liver Neoplasms; Stomach Neoplasms; Lymphoma | Details |
| RB-0004 | RB-0004 | Phase 1 Clinical | Reyoung Pharmaceutical Co Ltd | Solid tumours; Lymphoma | Details |
| Zeluvalimab | AMG-404 | Phase 1 Clinical | Amgen Inc | Solid tumours; Small Cell Lung Carcinoma; Prostatic Neoplasms, Castration-Resistant; Precursor Cell Lymphoblastic Leukemia-Lymphoma | Details |
| PF-07209960 | PF-07209960 | Phase 1 Clinical | Pfizer Inc | Squamous Cell Carcinoma of Head and Neck; Carcinoma, Renal Cell; Carcinoma, Transitional Cell; Carcinoma, Ovarian Epithelial; Colorectal Neoplasms; Carcinoma, Non-Small-Cell Lung | Details |
| EBV-specific TCR-T cell with anti-PD1 auto-secreted element | Phase 2 Clinical | Tcrcure Biopharma Ltd | Squamous Cell Carcinoma of Head and Neck | Details | |
| MK-3475A | MK-3475A | Phase 3 Clinical | Merck Sharp & Dohme Corp | Solid tumours; Ovarian Neoplasms; Carcinoma, Renal Cell; Lung Neoplasms; Carcinoma, Squamous Cell; Carcinoma, Non-Small-Cell Lung; Melanoma | Details |
| SPX-303 | SPX-303 | Phase 1 Clinical | SparX Biopharmaceutical Corp | Solid tumours | Details |
| INCA33890 | INCA33890; INCA-33890 | Phase 1 Clinical | Incyte Corp | Solid tumours; Neoplasms; Neoplasm Metastasis | Details |
| PD-1 knockout engineered T cells (Chengdu MedGenCell) | Phase 2 Clinical | West China Hospital Of Sichuan University, Chengdu Medgencell | Lung Neoplasms; Carcinoma, Non-Small-Cell Lung; Melanoma | Details | |
| PD-1 Inhibitor Therapy(Beijing YouAn Hospital) | Phase 2 Clinical | Beijing Youan Hospbeijing Youan Hospital,Capital Medical Universityital,Capital Medical University, Beijing Gene Key Life Technology Co Ltd | Carcinoma, Hepatocellular | Details | |
| CC-90006 | C-90006; CC-90006 | Phase 1 Clinical | Anaptysbio Inc | Autoimmune Diseases; Psoriasis | Details |
| NWY-001(Biocytogen Pharmaceuticals) | NWY001(Biocytogen Pharmaceuticals); YH008; YH-008; NWY-001(Biocytogen Pharmaceuticals) | Phase 1 Clinical | Eucure Pharmaceutical Technology (Beijing) Co Ltd, Biocytogen Pharmaceuticals (Beijing) Co Ltd | Solid tumours; Hematologic Neoplasms; Neoplasms | Details |
| PD-1 knockout engineered T cells (Anhui Kedgene Biotechnology) | Phase 2 Clinical | Anhui Korton Biological Technology Co Ltd, Hangzhou Cancer Hospital | Esophageal Neoplasms; Carcinoma, Hepatocellular | Details | |
| Nofazinlimab | CS-1003 | Phase 3 Clinical | Cstone Pharmaceuticals | Solid tumours; Neoplasms; Lymphoma; Carcinoma, Hepatocellular | Details |
| Taminadenant | NIR-178; PBF-509 | Phase 1 Clinical | Palobiofarma Sl | Solid tumours; Ovarian Neoplasms; Head and Neck Neoplasms; Carcinoma, Renal Cell; Carcinoma; Triple Negative Breast Neoplasms; Lymphoma, Large B-Cell, Diffuse; Neoplasms; Pancreatic Neoplasms; Prostatic Neoplasms, Castration-Resistant; Colorectal Neoplasms; Parkinson Disease; Carcinoma, Pancreatic Ductal; Carcinoma, Non-Small-Cell Lung; Melanoma | Details |
| dPD-1 hCD19 CART cell therapy (Innovative Cellular Therapeutics) | ICTCAR-014 | Phase 2 Clinical | Innovative Cellular Therapeutics Co Ltd | Lymphoma, Non-Hodgkin | Details |
| Multiple target antigen stimulating cell therapy (HengRui YuanZheng Biotechnology) | Phase 2 Clinical | Hengrui Yuanzheng (Shanghai) Biotechnology Co Ltd | Solid tumours; Liver Neoplasms; Stomach Neoplasms; Neoplasms; Liver Diseases | Details | |
| PE0105 | PE-0105 | Phase 1 Clinical | Shanghai Yunyi Health Technology Development Co Ltd | Solid tumours; Neoplasms | Details |
| PD-1 Inhibitor Therapy(Affiliated Hospital of Nantong University) | Phase 2 Clinical | Affiliated Hospital Of Nantong University | Esophageal Squamous Cell Carcinoma | Details | |
| SI-B003 | SI-B003 | Phase 2 Clinical | Sichuan Baili Pharmaceutical Co Ltd | Solid tumours; Stomach Neoplasms; Esophageal Neoplasms; Squamous Cell Carcinoma of Head and Neck; Carcinoma, Renal Cell; Carcinoma, Transitional Cell; Small Cell Lung Carcinoma; Triple Negative Breast Neoplasms; Breast Neoplasms; Colorectal Neoplasms; Carcinoma, Non-Small-Cell Lung; Uterine Cervical Neoplasms; Neoplasm Metastasis; Melanoma | Details |
| Pembrolizumab biosimilar (biocad) | BCD-201 | Phase 3 Clinical | Biocad | Solid tumours; Carcinoma, Non-Small-Cell Lung; Melanoma | Details |
| PD-1 inhibitor therapy(Shanghai Chest Hospital) | Phase 2 Clinical | Shanghai Chest Hospital | Esophageal Neoplasms; Esophageal Squamous Cell Carcinoma | Details | |
| Spartalizumab | PDR-001 | Phase 3 Clinical | Novartis Pharma Ag | Esophageal Squamous Cell Carcinoma; Chordoma; Digestive System Neoplasms; Neuroendocrine Tumors; Breast Neoplasms; Sarcoma; Prostatic Neoplasms; Cholangiocarcinoma; Nasopharyngeal Carcinoma; Colorectal Neoplasms; Gastrointestinal Stromal Tumors; Primary Myelofibrosis; Bone Marrow Diseases; Esophageal adenocarcinoma; Endometrial Neoplasms; Carcinoma, Squamous Cell; Microsatellite instability-high cancer; Carcinoma, Pancreatic Ductal; Lung Neoplasms; Sarcoma, Alveolar Soft Part; Leukemia, Myeloid, Acute; Thyroid Neoplasms; Metastatic breast cancer; Uterine Neoplasms; Lymphoma; Adenocarcinoma; Persistent Fetal Circulation Syndrome; Carcinoma, Hepatocellular; Carcinoma, Non-Small-Cell Lung; Melanoma; Uterine Cervical Neoplasms; Lymphoma, Large B-Cell, Diffuse; Solid tumours; Ovarian Neoplasms; Liver Neoplasms; Leukemia, Myeloid; Leukemia; Kidney Neoplasms; Preleukemia; Hematologic Diseases; Carcinoma, Renal Cell; Carcinoma; Stomach Neoplasms; Squamous Cell Carcinoma of Head and Neck; Anus Neoplasms; Triple Negati | Details |
| Tebotelimab | PD-1 X LAG-3; MGD-013 | Phase 3 Clinical | Macrogenics Inc | Triple Negative Breast Neoplasms; Adenocarcinoma; Carcinoma, Hepatocellular; Melanoma; Carcinoma, Non-Small-Cell Lung; Uterine Cervical Neoplasms; Neoplasm Metastasis; Endometrial Neoplasms; Cholangiocarcinoma; Neoplasms; Solid tumours; Small Cell Lung Carcinoma; Esophageal Neoplasms; Stomach Neoplasms; Squamous Cell Carcinoma of Head and Neck; Liver Neoplasms; Biliary Tract Neoplasms; Hematologic Neoplasms; Ovarian Neoplasms; Head and Neck Neoplasms | Details |
| Ezabenlimab | BI-754091 | Phase 2 Clinical | C.H. Boehringer Sohn Ag & Co. Kg | Sarcoma; Melanoma; Neoplasm Metastasis; Carcinoma, Non-Small-Cell Lung; Gastrointestinal Neoplasms; Carcinoma, Pancreatic Ductal; Carcinoma, Squamous Cell; Esophageal Squamous Cell Carcinoma; Carcinoma, Neuroendocrine; Lung Neoplasms; Colorectal Neoplasms; Head and Neck Neoplasms; Colonic Neoplasms; Small Cell Lung Carcinoma; Triple Negative Breast Neoplasms; Neoplasms; Pancreatic Neoplasms; Rectal Neoplasms; Squamous Cell Carcinoma of Head and Neck; Liver Neoplasms; Biliary Tract Neoplasms; Solid tumours | Details |
| SAR-445877 | SAR-445877; SAR445877 | Phase 2 Clinical | Sanofi | Solid tumours | Details |
| Autologous PD-1 antibody expressing mesothelin-targeted CAR-Tcells therapy (Shanghai Cell Therapy Research Institute) | Phase 2 Clinical | Shanghai Cell Therapy Research Institute | Solid tumours | Details | |
| Pembrolizumab/Vibostolimab | MK-7684A | Phase 3 Clinical | Merck & Co Inc | Urinary Bladder Neoplasms; Carcinoma, Hepatocellular; Uterine Cervical Neoplasms; Carcinoma, Non-Small-Cell Lung; Melanoma; Gallbladder Neoplasms; Endometrial Neoplasms; Lung Neoplasms; Cholangiocarcinoma; Ovarian Neoplasms; Neoplasms; Triple Negative Breast Neoplasms; Small Cell Lung Carcinoma; Stomach Neoplasms; Esophageal Neoplasms; Squamous Cell Carcinoma of Head and Neck; Hematologic Neoplasms | Details |
| Rilvegostomig | AZD-2936 | Phase 3 Clinical | Astrazeneca Plc | Biliary Tract Neoplasms; Solid tumours; Stomach Neoplasms; Esophageal Neoplasms; Bile Duct Neoplasms; Carcinoma, Non-Small-Cell Lung | Details |
| BAT-1308 | BAT-1308 | Phase 3 Clinical | Bio-Thera Solutions Ltd | Solid tumours; Endometrial Neoplasms; Uterine Cervical Neoplasms | Details |
| Vudalimab | XmAb-20717; XmAb-717; XmAb 717 | Phase 2 Clinical | Xencor Inc | Thymoma; Adenocarcinoma, Clear Cell; Cholangiocarcinoma; Nasopharyngeal Carcinoma; Breast Neoplasms; Vulvar Neoplasms; Prostatic Neoplasms; Astrocytoma; Colorectal Neoplasms; Endometrial Neoplasms; Microsatellite instability-high cancer; Carcinoma, Neuroendocrine; Carcinoma, Squamous Cell; Fallopian Tube Neoplasms; Thyroid Neoplasms; Uterine Cervical Neoplasms; Melanoma; Carcinoma, Hepatocellular; Carcinoma, Non-Small-Cell Lung; Carcinoma, Ovarian Epithelial; Ovarian Neoplasms; Solid tumours; Esophageal Neoplasms; Stomach Neoplasms; Carcinoma, Renal Cell; Carcinoma, Basal Cell; Squamous Cell Carcinoma of Head and Neck; Rectal Neoplasms; Biliary Tract Neoplasms; Carcinoma, Transitional Cell; Small Cell Lung Carcinoma; Hodgkin Disease; Colonic Neoplasms; Adenoma, Acidophil; Mesothelioma; Adnexal Diseases; Prostatic Neoplasms, Castration-Resistant | Details |
| BCMA-PD1 CAR T cell therapy (General Hospital of the People's Liberation Army) | Phase 2 Clinical | People'S Liberation Army General Hospital Military Service | Multiple Myeloma | Details | |
| BCD-217 | BCD-217 | Phase 3 Clinical | Biocad | Melanoma | Details |
| CD19-PD1-CAR T cell therapy (Chinese PLA General Hospital) | Phase 2 Clinical | Pla General Hospital | Lymphoma, B-Cell | Details | |
| Rulonilimab | F-520 | Phase 3 Clinical | Shandong New Time Pharmaceutical Co Ltd | Lymphoma, T-Cell, Peripheral; Solid tumours; Neoplasms; Carcinoma, Transitional Cell; Lymphoma, Large B-Cell, Diffuse; Carcinoma, Non-Small-Cell Lung; Carcinoma, Hepatocellular; Central Nervous System Lymphoma; Uterine Cervical Neoplasms | Details |
| 89Zr-N-sucDf-pembrolizumab | Phase 1 Clinical | Merck & Co Inc, Umcg The University Medical Center Groningen | Melanoma; Carcinoma, Non-Small-Cell Lung | Details | |
| CD-200-AR-L | CD-200-AR-L; hP-1-A-8 | Phase 1 Clinical | University Of Minnesota | Glioblastoma | Details |
| Peresolimab | LY-3462817 | Phase 2 Clinical | Eli Lilly And Company | Arthritis, Rheumatoid; Autoimmune Diseases; Connective Tissue Diseases; Immune System Diseases; Joint Diseases; Psoriasis; Musculoskeletal Diseases; Rheumatic Diseases; Arthritis | Details |
| Latikafusp | AMG-256 | Phase 1 Clinical | Amgen Inc | Solid tumours | Details |
| Recombinant humanized anti-PD-1 monoclonal antibody (Anhui Anke Biotechnology) | Phase 2 Clinical | Anhui Anke Biotechnology (Group) Co Ltd | Esophageal Neoplasms; Carcinoma, Transitional Cell; Triple Negative Breast Neoplasms; Neoplasms; Lymphoma; Lung Neoplasms | Details | |
| Budigalimab | ABBV-181 | Phase 3 Clinical | Abbvie Inc | HIV Infections; Solid tumours; Head and Neck Neoplasms; Squamous Cell Carcinoma of Head and Neck; Neoplasms; Pancreatic Neoplasms; Small Cell Lung Carcinoma; Triple Negative Breast Neoplasms; Carcinoma, Hepatocellular; Carcinoma, Non-Small-Cell Lung | Details |
| Lipustobart | LZM-009 | Phase 2 Clinical | Livzon(Group) Pharmaceutical Factory | Solid tumours; Thymoma; Carcinoma, Non-Small-Cell Lung | Details |
| ABBV-1882 | ABBV-1882 | Phase 1 Clinical | Abbvie Inc | HIV Infections | Details |
| Pidilizumab | MDV-9300; CT-011 | Phase 2 Clinical | Curetech Ltd | Ovarian Neoplasms; Carcinoma, Renal Cell; Lymphoma, Large B-Cell, Diffuse; Pancreatic Neoplasms; Colonic Neoplasms; Multiple Myeloma; Prostatic Neoplasms; Breast Neoplasms; Sarcoma; Colorectal Neoplasms; Lymphoma; Lymphoma, Non-Hodgkin; Hepatitis C, Chronic; Melanoma; Carcinoma, Hepatocellular | Details |
| MAX-10181 | MAX-10181; MAX-1 | Phase 2 Clinical | Maxinovel Pharmaceuticals Co Ltd | Solid tumours; Neoplasms | Details |
| Nivolumab biosimilar(Amgen) | ABP-206 | Phase 3 Clinical | Amgen Inc | Melanoma | Details |
| MEDI-0680 | AMP-514; MEDI-0680 | Phase 2 Clinical | Medimmune | Lymphoma, B-Cell; Kidney Neoplasms; Carcinoma, Renal Cell; Neoplasms | Details |
| Acrixolimab | YBL-006; YBL 006 | Phase 2 Clinical | Y-biologics Inc | Solid tumours | Details |
| RO-7284755 | RO-7284755 | Phase 1 Clinical | F. Hoffmann-La Roche Ag | Solid tumours; Neoplasms | Details |
| MiHA-loaded PD-L silenced DC vaccination (Radboud University) | Phase 2 Clinical | Radboud University Nijmegen | Hematologic Neoplasms; Colorectal Neoplasms | Details | |
| Sym-021 | Sym-021 | Phase 1 Clinical | Symphogen A/S | Solid tumours; Lymphoma; Neoplasm Metastasis | Details |
| SHR-1901 | SHR-1901 | Phase 1 Clinical | Suzhou Suncadia Biopharmaceuticals Co Ltd | Neoplasms | Details |
| TQB-2868 | TQB-2868 | Phase 2 Clinical | Nanjing Shunxin Pharmaceuticals Co Ltd Of Chiatai Tianqing Pharmaceutical Group | Neoplasms; Uterine Cervical Neoplasms; Carcinoma, Hepatocellular | Details |
| Mavezelimab/Pembrolizumab | MK-4280A; MK-4820A | Phase 3 Clinical | Msd Ireland (Carlow) Merck Sharp & Dohme Corp, MSD R&D(China)Co Ltd | Solid tumours; Carcinoma, Renal Cell; Hodgkin Disease; Carcinoma, Transitional Cell; Small Cell Lung Carcinoma; Colorectal Neoplasms; Carcinoma, Squamous Cell; Endometrial Neoplasms | Details |
| BA1104 | LY-01015; BA-1104 | Phase 3 Clinical | Luye Pharma Group Ltd | Liver Neoplasms; Squamous Cell Carcinoma of Head and Neck; Esophageal Neoplasms; Stomach Neoplasms; Carcinoma, Renal Cell; Mesothelioma; Urinary Bladder Neoplasms; Glioma; Esophageal Squamous Cell Carcinoma; Carcinoma, Non-Small-Cell Lung; Melanoma | Details |
| Genolimzumab | GB-226; APL-501; APL501; CBT-501; GB226 | Phase 2 Clinical | Cbt, Genor Biopharma Co Ltd, Apollomics Inc | Colorectal Neoplasms; Carcinoma, Non-Small-Cell Lung; Carcinoma, Hepatocellular; Uterine Cervical Neoplasms; Neoplasms, Unknown Primary; Lymphoma, Non-Hodgkin; Lymphoma; Sarcoma, Alveolar Soft Part; Thymoma; Lymphoma, T-Cell, Peripheral; Thymus Neoplasms; Microsatellite Instability; Hodgkin Disease; Carcinoma, Renal Cell; Mismatch Repair Deficient Cancer; Lymphoma, B-Cell; Solid tumours | Details |
| Lorigerlimab | MGD-019; AEX1344 | Phase 2 Clinical | Macrogenics Inc | Solid tumours; Liver Neoplasms; Skin Melanoma; Carcinoma, Renal Cell; Carcinoma, Ovarian Epithelial; Neoplasms; Prostatic Neoplasms, Castration-Resistant; Prostatic Neoplasms; Colorectal Neoplasms; Carcinoma, Squamous Cell; Carcinoma, Pancreatic Ductal; Uterine Cervical Neoplasms; Neoplasm Metastasis; Melanoma; Carcinoma, Non-Small-Cell Lung | Details |
| Sym-024 | Sym-024; S-95024 | Phase 1 Clinical | Symphogen A/S | Solid tumours; Neoplasm Metastasis | Details |
| Recombinant human anti-PD-1 monoclonal antibody(Hec Biological) | Phase 1 Clinical | Dongguan City Hec Biological Medicine Res And Development Co Ltd | Stomach Neoplasms; Esophageal adenocarcinoma | Details | |
| T3011 | MVR-T3011; T3011; MVR-T3011 IT; MVR-T3011 IV | Phase 2 Clinical | Immvira Co Ltd | Sarcoma; Carcinoma, Non-Small-Cell Lung; Carcinoma, Hepatocellular; Melanoma; Carcinoma, Squamous Cell; Lymphoma; Lung Neoplasms; Endometrial Neoplasms; Colorectal Neoplasms; Ovarian Neoplasms; Breast Neoplasms; Mesothelioma; Skin Neoplasms; Squamous Cell Carcinoma of Head and Neck; Esophageal Neoplasms; Solid tumours; Head and Neck Neoplasms; Liver Neoplasms | Details |
| GX-P1 | GX-P1 | Phase 1 Clinical | Genexine Inc | Autoimmune Diseases | Details |
| EMB-02 | EMB-02 | Phase 2 Clinical | Shanghai Epimab Biotherapeutics, Inc | Solid tumours | Details |
| PD-1 knockout EBV-CTL (Nanjing Medical University) | Phase 2 Clinical | Nanjing Medical University | Stomach Neoplasms; Hodgkin Disease; Lymphoma, Large B-Cell, Diffuse; Nasopharyngeal Neoplasms; Lymphoma | Details | |
| Recombinant human PD-1 antibody herpes simplex virus | Phase 1 Clinical | Yangsheng Tang Co Ltd, Xiamen University | Liver Neoplasms; Solid tumours; Head and Neck Neoplasms; Neoplasms; Pancreatic Neoplasms; Urinary Bladder Neoplasms; Central Nervous System Neoplasms; Colorectal Neoplasms; Brain metastases; Lung Neoplasms; Melanoma; Uterine Cervical Neoplasms | Details | |
| IMU-201 | IMU-201 | Phase 1 Clinical | Imugene Ltd | Adenocarcinoma of Lung; Carcinoma, Large Cell; Carcinoma, Squamous Cell; Lung Neoplasms; Carcinoma, Non-Small-Cell Lung | Details |
| HerinCAR-PD1 | Phase 2 Clinical | Ningbo Cancer Hospital | Solid tumours | Details | |
| CD19 PD-1/CD28 CAR-T Cell Therapy (Second Affiliated Hospital School Of Zhejiang University School Of Medicine) | Phase 1 Clinical | Second Affiliated Hospital Of Zhejiang University School Of Medicine | Lymphoma, B-Cell; Lymphoma, Large B-Cell, Diffuse; Lymphoma, Mantle-Cell; Lymphoma, Follicular; Primary mediastinal B cell lymphoma | Details | |
| PH-762 | PH-762; PH762-ACT; RXI 762-ACT | Phase 1 Clinical | Phio Pharmaceuticals Corp | Skin Melanoma; Squamous Cell Carcinoma of Head and Neck; Skin Neoplasms; Breast Neoplasms; Genital Neoplasms, Female; Colorectal Neoplasms; Carcinoma, Squamous Cell; Urogenital Neoplasms; Lung Neoplasms; Melanoma | Details |
| AK-129 | AK-129 | Phase 1 Clinical | Zhongshan Akeso Biopharma Co Ltd | Neoplasms | Details |
| Fidasimtamab | IBI-315 | Phase 1 Clinical | Hanmi Pharmaceutical Co Ltd, Innovent Biologics(Suzhou) Co Ltd | Solid tumours; Neoplasms | Details |
| STW204 | STW-204; AI025; AI-025 | Phase 1 Clinical | Suzhou Stainwei Biotech Inc | Solid tumours; Neoplasms | Details |
| ZG-005 | ZG-005 | Phase 2 Clinical | Suzhou Zelgen Biopharmaceuticals Co Ltd | Solid tumours; Carcinoma, Neuroendocrine; Uterine Cervical Neoplasms | Details |
| STW204/Gotistobart | AI-061 | Phase 1 Clinical | Oncoc4 Inc | Bile Duct Neoplasms; Uterine Cervical Neoplasms; Carcinoma, Non-Small-Cell Lung; Gastrointestinal Neoplasms; Melanoma; Carcinoma, Hepatocellular; Endometrial Neoplasms; Fallopian Tube Neoplasms; Colorectal Neoplasms; Carcinoma, Renal Cell; Peritoneal Neoplasms; Urinary Bladder Neoplasms; Digestive System Neoplasms; Stomach Neoplasms; Anus Neoplasms; Esophageal Neoplasms; Squamous Cell Carcinoma of Head and Neck; Cystadenocarcinoma, Serous | Details |
| KY-0118 | KY-0118; KY0118 | Phase 1 Clinical | Novatim Immune Therapeutics (Zhejiang) Co Ltd | Solid tumours; Carcinoma, Renal Cell; Carcinoma, Transitional Cell; Neoplasms; Pancreatic Neoplasms; Colorectal Neoplasms; Melanoma; Carcinoma, Non-Small-Cell Lung | Details |
| JNJ-67484703 | JNJ-67484703; JNJ-4703 | Phase 1 Clinical | Janssen Research & Development Llc | Arthritis, Rheumatoid | Details |
| INSIX RA (Indus Biotech) | Clinical | Indus Biotech Pvt Ltd | Arthritis, Rheumatoid; Inflammation | Details | |
| VT1093 | VT1093; VT-1093 | Phase 1 Clinical | Beijing Weiyuan Likang Biotechnology Co Ltd | Solid tumours | Details |
| CMAB-819 | CMAB8-19 | Phase 1 Clinical | Sinomab Bioscience Ltd | Squamous Cell Carcinoma of Head and Neck; Carcinoma, Hepatocellular; Carcinoma, Non-Small-Cell Lung | Details |
| BCD-263 | BCD-263 | Phase 1 Clinical | Biocad | Melanoma | Details |
| CDX-585 | CDX-585 | Phase 1 Clinical | Celldex Therapeutics | Liver Neoplasms; Ovarian Neoplasms; Head and Neck Neoplasms; Solid tumours; Carcinoma, Renal Cell; Esophageal Neoplasms; Stomach Neoplasms; Neoplasms; Pancreatic Neoplasms; Urinary Bladder Neoplasms; Cholangiocarcinoma; Colorectal Neoplasms; Peritoneal Neoplasms; Fallopian Tube Neoplasms; Carcinoma, Non-Small-Cell Lung | Details |
| Recombinant humanized anti-PD-1 monoclonal antibody (Shanxi Wichida Bright Pharmaceutical) | Phase 1 Clinical | Shanxi Weiqidaguangming Pharmaceutical Co Ltd | Solid tumours | Details | |
| MW-11 | MW-11; 9MW1111; 9-MW1111; 9MW-1111; 9-MW-1111 | Phase 1 Clinical | Mabwell (Shanghai) Bioscience Co Ltd | Solid tumours; Breast Neoplasms | Details |
| Reozalimab | IBI-318; LY-3434172; LY3434172 | Phase 1 Clinical | Innovent Biologics(Suzhou) Co Ltd, Eli Lilly And Company | Neoplasms; Small Cell Lung Carcinoma; Lymphoma, Extranodal NK-T-Cell; Carcinoma, Squamous Cell; Carcinoma, Hepatocellular | Details |
| GNR-051 | GNR-051 | Phase 1 Clinical | Generium Pharmaceuticals | Carcinoma, Renal Cell; Melanoma; Carcinoma, Non-Small-Cell Lung | Details |
| SOT-201 | SOT-201 | Phase 1 Clinical | Sotio | Solid tumours; Neoplasms; Neoplasm Metastasis | Details |
| AZD-7789 | AZD-7789 | Phase 2 Clinical | AstraZeneca Ag | Esophageal Neoplasms; Stomach Neoplasms; Hodgkin Disease; Digestive System Neoplasms; Carcinoma, Non-Small-Cell Lung; Gastrointestinal Neoplasms | Details |
| Pegylated recombinant human granulocyte colony stimulating factor(Sun Yat-sen University) | Phase 1 Clinical | Sun Yat-Sen University | Hematologic Neoplasms; Nasopharyngeal Carcinoma | Details | |
| IBI-319 | IBI-319 | Phase 1 Clinical | Innovent Biologics(Suzhou) Co Ltd | Neoplasms | Details |
| Sasanlimab | PF-6801591; PF-06801591; RN-888 | Phase 3 Clinical | Pfizer Pharmaceuticals Ltd (China), Pfizer Inc | Urinary Bladder Neoplasms; Fructose Intolerance; Gastrointestinal Neoplasms; Carcinoma, Non-Small-Cell Lung; Adenocarcinoma; Melanoma; Urogenital Neoplasms; Endometrial Neoplasms; Carcinoma, Squamous Cell; Bile Duct Neoplasms; Colorectal Neoplasms; Urologic Neoplasms; Prostatic Neoplasms; Sarcoma; Urologic Diseases; Digestive System Neoplasms; Kidney Neoplasms; Kidney Diseases; Carcinoma, Transitional Cell; Pancreatic Neoplasms; Neoplasms; Neoplasms, Glandular and Epithelial; Squamous Cell Carcinoma of Head and Neck; Esophageal Neoplasms; Stomach Neoplasms; Carcinoma, Renal Cell; Carcinoma; Ovarian Neoplasms; Solid tumours; Head and Neck Neoplasms; Liver Neoplasms | Details |
| Treprilimab | Clinical | Sun Yat-Sen University | Oropharyngeal Neoplasms | Details | |
| Cetrelimab | JNJ-63723283; JNJ-3283 | Phase 3 Clinical | Johnson & Johnson | Hepatitis B, Chronic; Neoplasms; Carcinoma, Transitional Cell; Pancreatic Neoplasms; Urinary Bladder Neoplasms; Multiple Myeloma; Prostatic Neoplasms, Castration-Resistant; Prostatic Neoplasms; Carcinoma, Neuroendocrine; Carcinoma, Small Cell; Melanoma; Carcinoma, Non-Small-Cell Lung | Details |
| Rituximab/Paclitaxel | AR-160 | Phase 1 Clinical | Mayo Clinic | Lymphoma, Non-Hodgkin; Leukemia, Lymphocytic, Chronic, B-Cell | Details |
| Mableukin 2PD1(Anwita Biosciences) | AWT020; AWT-020 | Phase 1 Clinical | Anwita Biosciences Inc | Neoplasms; Autoimmune Diseases of the Nervous System | Details |
| Pembrolizumab/Quavonlimab | MK-1308A | Phase 3 Clinical | Merck Sharp & Dohme Corp, Msd Ireland (Carlow), MSD R&D(China)Co Ltd | Solid tumours; Carcinoma, Renal Cell; Small Cell Lung Carcinoma; Colorectal Neoplasms; Carcinoma, Hepatocellular; Melanoma | Details |
| JS-207 | JS-207 | Phase 1 Clinical | Shanghai Junshi Biological Engineering Co Ltd | Neoplasms | Details |
| Anti-PD-1 Antibody(Fujian Cancer Hospital) | Phase 2 Clinical | Carcinoma, Hepatocellular | Details | ||
| 64Cu-DOTA-nivolumab (Bristol-Myers Squibb/Ono Pharmaceutical) | Clinical | Ono Pharmaceutical Co Ltd, Bristol-Myers Squibb Company | Melanoma; Carcinoma, Non-Small-Cell Lung | Details | |
| IBI-363 | IBI-363 | Phase 2 Clinical | Innovent Biologics(Suzhou) Co Ltd | Solid tumours; Neoplasms; Lymphoma; Melanoma | Details |
| αPD1-MSLN-CAR T cell therapy (Shanghai Cell Therapy Group Co.,Ltd) | BZDS1901; BZDS-1901; BZD1901 | Phase 1 Clinical | Shanghai Cell Therapy Group Co Ltd | Ovarian Neoplasms; Solid tumours; Mesothelioma; Colorectal Neoplasms; Carcinoma, Non-Small-Cell Lung | Details |
| AK-131 | AK-131 | Phase 1 Clinical | Zhongshan Akeso Biopharma Co Ltd | Solid tumours; Neoplasms | Details |
| Volrustomig | MEDI-5752; MEDI5752 | Phase 3 Clinical | Medimmune Llc | Biliary Tract Neoplasms; Solid tumours; Stomach Neoplasms; Esophageal Neoplasms; Squamous Cell Carcinoma of Head and Neck; Carcinoma, Renal Cell; Mesothelioma; Sarcoma; Bile Duct Neoplasms; Carcinoma, Hepatocellular; Uterine Cervical Neoplasms; Carcinoma, Non-Small-Cell Lung | Details |
This web search service is supported by Google Inc.





